The Significance of Vasospasm in Stable Coronary Artery Disease
The clinical hallmark of stable coronary artery disease is angina pectoris caused by physical or emotional stress that is relieved by rest or nitroglycerine intake. In addition, many patients with suspected coronary artery disease report angina pectoris at rest and the Canadian Cardiovascular Society Classification for stable angina states that resting angina may occur at every class together with angina on exertion.1 Despite signs and symptoms of myocardial ischemia convincing enough for a cardiologist to refer the patient for invasive diagnostic coronary angiography, a substantial number of these patients does not have a relevant stenosis. In case of an intermediate stenosis assessment with FFR is indicated to determine the relevance of a lesion; however, in case of a non-hemodynamically relevant stenosis or even no stenosis at all the question remains whether or not the patient's symptoms are of cardiac origin. This scenario is not infrequent as more than 50% of patients with suspected coronary artery disease are found to have unobstructed coronary arteries when coronary angiography is performed.2 One may argue that patient selection and non-invasive ischemia testing needs to be improved in order to enhance the number of patients with significant epicardial stenosis. However, recent studies have shown that even in the presence of an abnormal non-invasive imaging stress test the frequency of unobstructed coronary arteries on diagnostic coronary angiography was 54%.3 Moreover, a negative non-invasive stress test does not rule out the possibility of a functional coronary vasomotor disorder as a cardiac cause for the clinical presentation.4
It is well documented that patients with chest pain and unobstructed coronary arteries have a high morbidity with more depression and anxiety, premature loss of workforce and more need for antianginal medication causing higher costs to the healthcare systems than patients with obstructive coronary artery disease.5,6,7 Moreover, recent studies have suggested that this group of patients has an increased risk of approximately 1,7%/year for myocardial infarction or death which is significantly higher than in a matched healthy cohort.8
As such patients are often discharged with a diagnosis of non-cardiac chest pain and hence without any anti-anginal medication, it is important to recognize that functional coronary disorders can be responsible for a presentation of stable angina not due to an epicardial stenosis (it is assumed that obvious cardiac diseases such as hypertrophic cardiomyopathy, aortic valve disease, congenital heart disease et cetera were ruled out during the initial work-up of the patient).
One of the functional abnormalities causing stable angina despite unobstructed coronary arteries is coronary artery spasm which can occur at the epicardial level as well as in the coronary microcirculation.9 Although epicardial coronary artery spasm is frequently associated with angina pectoris at rest, it has been also described in patients with purely exercise-related symptoms.10,11,12 Vasomotion disorders of the small coronary vessels were demonstrated more than 20 years ago by Pupita et al. who described findings from ambulatory ECG recordings and exercise testing following intravenous ergonovine application in patients with a chronic total occlusion who had no other coronary lesions.13 They showed that ischemia occurred earlier following ergonovine administration indicating that ergonovine-induced constriction of peripheral coronary arteries or collaterals was responsible for this observation. When these patients had coronary angiography following intracoronary ergonovine administration constriction of collateral vessels associated with angina and ST-segment depression could be demonstrated in some patients. This suggests that vasoconstriction of small coronary vessels modulates the angina threshold and may be solely responsible for the occurrence of anginal symptoms during exercise.
A tendency for coronary vasospasm seems to be rather common in patients with chronic stable angina irrespective of the degree of epicardial narrowing suggesting that functional coronary abnormalities may be the only cause of angina but may also be superimposed in patients who have significant coronary narrowings (≥50%). Vasospasm was common in patients with no or only mild epicardial coronary irregularities enrolled in the ACOVA study. Of the 376 European patients with stable angina and suspected coronary artery disease 47% had no epicardial stenosis (≤20% narrowings).14 Of these, 86% underwent intracoronary acetylcholine (ACH) testing and 28% had epicardial and 34% microvascular spasm. Of note, women were more likely to have unobstructed coronary arteries and they were more often found to have a pathologic ACH-test. A positive family history for cardiovascular disease was more often present in patients with abnormal coronary vasomotion compared to those with an uneventful ACH-test. A recent study from Japan reported a similar frequency of 57% of pathologic ACH-tests (88% epicardial, 12% microvascular) among 1760 patients with typical or atypical angina-like chest pain who were referred for invasive diagnostic coronary angiography.15 This study also included patients (approximately 20%) with an epicardial stenosis of >75% indicating that an impaired coronary vasomotion also contributes to symptoms in patients with relevant epicardial narrowings.
Another group of patients with stable angina in whom coronary artery spasm has been described as being responsible for the clinical presentation are those with ongoing or recurrent angina despite successful stent implantation. Several studies in Asian and Caucasian patients have shown that coronary spasm occurs in a diffuse manner distal to the stent in approximately 70% of patients assessed.16,17,18 The underlying mechanisms are still incompletely understood and there is currently an ongoing debate whether or not the drug coating of drug eluting stents may facilitate such a pathologic vasoreaction.19
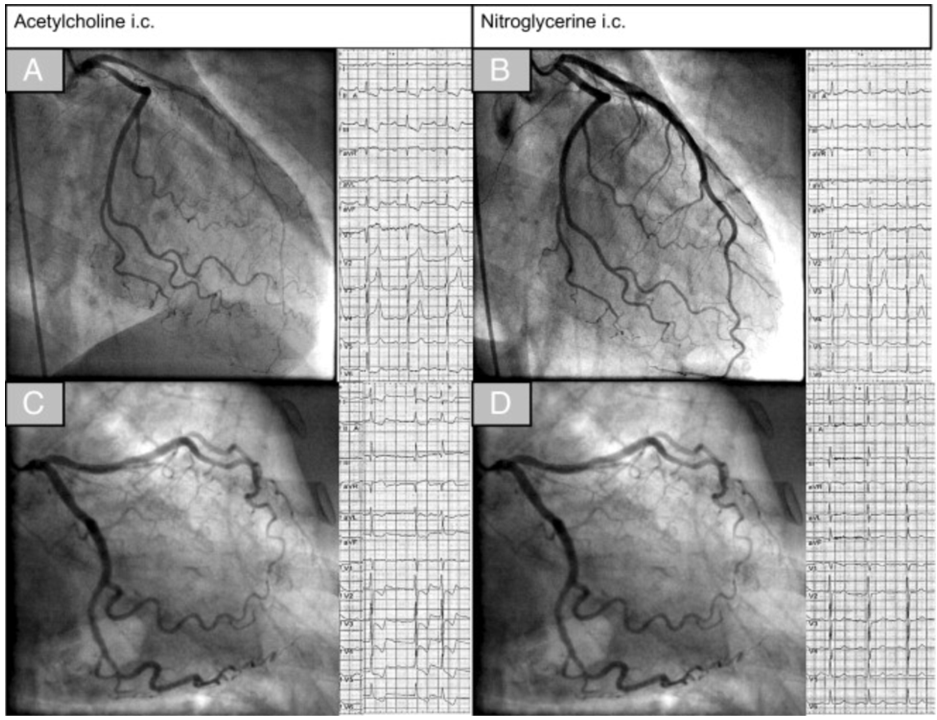
Coronary artery spasm can be diagnosed e.g. using intracoronary acetylcholine provocation testing that can be performed immediately following diagnostic coronary angiography. Standardized testing protocols have been developed in the past decades in specialized institutions using incremental doses of 2µg, 20µg, 100µg and up to 200µg for assessment of the left coronary artery and 50µg, 80µg in the right coronary artery. Continuous 12-lead-ECG registration during the test is mandatory and the patient should be interrogated about any symptoms occurring during the test after each step. Epicardial spasm is usually defined as reproduction of the patient's symptoms, ischemic ECG shifts and epicardial vasoconstriction >90% during acetylcholine provocation (Figure 1A+B).8 Microvascular spasm is defined as reproduction of the patient's symptoms, ischemic ECG shifts but no relevant epicardial vasoconstriction (Figure 1C+D).20 A detailed description of the diagnostic tools for assessment of coronary spasm and its interpretation can be found elsewhere.21,22 It is important to remember that such invasive coronary reactivity testing is safe with a reported complication rate of around 1% 23,24 which is comparable to the complication rate of invasive diagnostic coronary angiography.25,26 Once the diagnosis is established, patients should be treated with calcium channel blockers and short acting nitrates in addition to cardiovascular prevention drugs in accordance with current guidelines. We hope to encourage interventional cardiologists to implement intracoronary reactivity testing for the assessment of coronary artery spasm in their daily routine. Multicenter international studies may be useful to better understand the complex interplay between anatomic and functional coronary artery disease and their relation to angina symptoms. Such studies will be conducted by the Coronary Vasomotor Disorders International Study Group (COVADIS)27 with the intention to address current knowledge gaps regarding pharmacological treatment, ethnic and gender differences as well as prognosis.
Figure 1
The upper panels show left coronary artery angiograms and electrocardiograms (ECGs) in a patient with epicardial spasm. Note the diffuse but distally accentuated narrowing of the left anterior descending coronary artery (wraps around the apex) during acetylcholine infusion together with ischemic ECG shifts in the inferior leads (A) and resolution of both findings after intracoronary nitroglycerin (B). The lower panels show an example of a patient with microvascular spasm. During acetylcholine infusion the patient had reproduction of chest pain and ischemic ECG changes but no epicardial vasoconstriction (C). After intracoronary nitroglycerin, chest pain and ECG changes resolved (D). From: High Prevalence of a Pathological Response to Acetylcholine Testing in Patients With Stable Angina Pectoris and Unobstructed Coronary Arteries: The ACOVA Study (Abnormal COronary VAsomotion in patients with stable angina and unobstructed coronary arteries) J Am Coll Cardiol 2012;59:655-662.
References
- Campeau L. The Canadian Cardiovascular Society grading of angina pectoris revisited 30 years later. Can J Cardiol 2002;18:371-9.
- Patel MR, Peterson ED, Dai D, Brennan JM, Redberg RF, Anderson HV, Brindis RG, Douglas PS. Low diagnostic yield of elective coronary angiography. N Engl J Med 2010;362:886-95.
- Neglia D, Rovai D, Caselli C, et al.. Detection of significant coronary artery disease by noninvasive anatomical and functional imaging. Circ Cardiovasc Imaging 2015;8,pii:e002179.
- Lack of correlation between noninvasive stress tests and invasive coronary vasomotor dysfunction in patients with nonobstructive coronary artery disease. Cassar A, Chareonthaitawee P, Rihal CS, Prasad A, Lennon RJ, Lerman LO, Lerman A. Circ Cardiovasc Interv 2009;2:237-44.
- Jespersen L, Abildstrøm SZ, Hvelplund A, Prescott E. Persistent angina: highly prevalent and associated with long-term anxiety, depression, low physical functioning, and quality of life in stable angina pectoris. Clin Res Cardiol 2013;102:571-81.
- Jespersen L, Abildstrøm SZ, Hvelplund A, Galatius S, Madsen JK, Pedersen F, Højberg S, Prescott E. Symptoms of angina pectoris increase the probability of disability pension and premature exit from the workforce even in the absence of obstructive coronary artery disease. Eur Heart J 2013;34:3294-303.
- Shaw LJ, Merz CN, Pepine CJ, Reis SE, Bittner V, Kip KE, Kelsey SF, Olson M, Johnson BD, Mankad S, Sharaf BL, Rogers WJ, Pohost GM, Sopko G; Women's Ischemia Syndrome Evaluation (WISE) Investigators. The economic burden of angina in women with suspected ischemic heart disease: results from the National Institutes of Health–National Heart, Lung, and Blood Institute–sponsored Women's Ischemia Syndrome Evaluation. Circulation 2006;114:894–904.
- Jespersen L, Hvelplund A, Abildstrøm SZ, Pedersen F, Galatius S, Madsen JK, Jørgensen E, Kelbæk H, Prescott E. Stable angina pectoris with no obstructive coronary artery disease is associated with increased risks of major adverse cardiovascular events. Eur Heart J 2012;33:734-44.
- JCS Joint Working Group. Guidelines for diagnosis and treatment of patients with vasospastic angina (Coronary Spastic Angina) (JCS 2013). Circ J. 2014;78:2779-2801.
- Boden WE, Bough EW, Korr KS, Benham I, Gheorghiade M, Caputi A, Shulman RS. Exercise-induced coronary spasm with S-T segment depression and normal coronary arteriography. Am J Cardiol 1981;48:193-7.
- Yilmaz A, Hill S, Schäufele T, Vöhringer M, Geissler A, Sechtem U. Exercise-induced spastic coronary artery occlusion at the site of a moderate stenosis: neither Prinzmetal's angina nor cardiac syndrome X but "Prinzmetal X". Circulation 2010;122:e570–4.
- Miwa K, Fujita M, Ejiri M, Sasayama S. Comparative sensitivity of intracoronary injection of acetylcholine for the induction of coronary spasm in patients with various types of angina pectoris. Am Heart J 1990;120:544-50.
- Pupita G, Maseri A, Kaski JC, Galassi AR, Gavrielides S, Davies G, Crea F. Myocardial ischemia caused by distal coronary-artery constriction in stable angina pectoris. N Engl J Med 1990;323:514-20.
- Ong P, Athanasiadis A, Borgulya G, Mahrholdt H, Kaski JC, Sechtem U. High prevalence of a pathological response to acetylcholine testing in patients with stable angina pectoris and unobstructed coronary arteries. The ACOVA Study (Abnormal COronary VAsomotion in patients with stable angina and unobstructed coronary arteries). J Am Coll Cardiol 2012;59:655-62.
- Sato K, Kaikita K, Nakayama N, Horio E, Yoshimura H, Ono T, Ohba K, Tsujita K, Kojima S, Tayama S, Hokimoto S, Matsui K, Sugiyama S, Yamabe H, Ogawa H. Coronary vasomotor response to intracoronary acetylcholine injection, clinical features, and long-term prognosis in 873 consecutive patients with coronary spasm: analysis of a single-center study over 20 years. J Am Heart Assoc 2013;2:e000227.
- Lim SH, Flammer AJ, Yoon MH, Lennon RJ, Gulati R, Mathew V, Rihal CS, Lerman LO, Lerman A. The long-term effect of coronary stenting on epicardial and microvascular endothelial function. Circ Cardiovasc Interv 2012;5:523-9.
- Ong P, Athanasiadis A, Perne A, Mahrholdt H, Schäufele T, Hill S, Sechtem U. Coronary vasomotor abnormalities in patients with stable angina after successful stent implantation but without in-stent restenosis. Clin Res Cardiol 2014;103:11-19.
- Ito S, Nakasuka K, Morimoto K, Inomata M, Yoshida T, Tamai N, Suzuki S, Murakami Y, Morino A, Shimizu Y, Sato K. Angiographic and clinical characteristics of patients with acetylcholine-induced coronary vasospasm on follow-up coronary angiography following drug-eluting stent implantation. J Invasive Cardiol 2011;23:57-64.
- Nakazawa G, Shinke T, Ijichi T, Matsumoto D, Otake H, Torii S, Hiranuma N, Ohsue T, Otsuka F, Shite J, Hirata K, Ikari Y. Comparison of vascular response between durable and biodegradable polymer-based drug-eluting stents in a porcine coronary artery model. EuroIntervention 2014;10:717-23.
- Mohri M, Koyanagi M, Egashira K, Tagawa H, Ichiki T, Shimokawa H, Takeshita A. Angina pectoris caused by coronary microvascular spasm. Lancet 1998;351:1165-9.
- Ong P, Athanasiadis A, Sechtem U. Patterns of coronary vasomotor responses to intracoronary acetylcholine provocation. Heart 2013;99:1288-95.
- Zaya M, Mehta PK, Merz CN. Provocative testing for coronary reactivity and spasm. J Am Coll Cardiol 2014;63:103-9.
- Wei J, Mehta PK, Johnson BD, Samuels B, Kar S, Anderson RD, Azarbal B, Petersen J, Sharaf B, Handberg E, Shufelt C, Kothawade K, Sopko G, Lerman A, Shaw L, Kelsey SF, Pepine CJ, Merz CN. Safety of coronary reactivity testing in women with no obstructive coronary artery disease: results from the NHLBI-sponsored WISE (Women's Ischemia Syndrome Evaluation) study. JACC Cardiovasc Interv. 2012;5:646-53.
- Ong P, Athanasiadis A, Borgulya G, Vokshi I, Bastiaenen R, Kubik S, Hill S, Schäufele T, Mahrholdt H, Kaski JC, Sechtem U. Clinical usefulness, angiographic characteristics, and safety evaluation of intracoronary acetylcholine provocation testing among 921 consecutive white patients with unobstructed coronary arteries. Circulation 2014;129:1723-30.
- Noto TJ Jr, Johnson LW, Krone R, Weaver WF, Clark DA, Kramer JR Jr, Vetrovec GW. Cardiac catheterization 1990: a report of the Registry of the Society for Cardiac Angiography and Interventions (SCA&I). Cathet Cardiovasc Diagn 1991;24:75–83.
- Chandrasekar B, Doucet S, Bilodeau L, Crepeau J, deGuise P, Gregoire J, Gallo R, Cote G, Bonan R, Joyal M, Gosselin G, Tanguay JF, Dyrda I, Bois M, Pasternac A. Complications of cardiac catheterization in the current era: a single-center experience. Catheter Cardiovasc Interv 2001;52:289–295.
- Beltrame JF, Crea F, Kaski JC, Ogawa H, Ong P, Sechtem U, Shimokawa H, Baiery Merz CN. On Behalf of the Coronary Vasomotion Disorders International Study Group (COVADIS). International standardization of diagnostic criteria for vasospastic angina. Eur Heart J 2015. doi:10.1093/eurheartj/ehv351
Keywords: Acetylcholine, Angina Pectoris, Angina, Stable, Angina, Unstable, Anxiety, Calcium Channel Blockers, Cardiomyopathy, Hypertrophic, Cardiovascular Agents, Constriction, Constriction, Pathologic, Coronary Angiography, Coronary Artery Disease, Coronary Vasospasm, Drug-Eluting Stents, Electrocardiography, Ergonovine, Heart Valve Diseases, Microcirculation, Myocardial Infarction, Nitrates, Nitroglycerin, Patient Selection, Physical Exertion, Pipecolic Acids, Prevalence, Spasm, Stress, Psychological, Vasoconstriction
< Back to Listings