How to Mend a Broken Heart: An Approach to the Failing RV
Introduction
Right ventricular (RV) failure is a recognized complication of primary cardiac and pulmonary vascular disorders and is associated with a poor prognosis. Understanding RV physiology is paramount for adequate management of patients presenting with RV failure.
RV Preload and Ejection
The RV ejects into a low-resistance, high-compliance pulmonary vascular bed. It can sustain a significant increase in preload without increase in filling pressure.1 In normal conditions, RV and left ventricular (LV) stroke volume are equal; however, RV end diastolic volume is slightly bigger. Hence, ejection fraction of a normal RV is lower than that of the LV (normal RV ejection fraction is above 45%).2
Ventriculo-Arterial Coupling
To function efficiently in case of elevation of pulmonary vascular resistance (PVR), the RV will adapt its elastance (end systolic pressure divided by end systolic volume) to match the elastance of the arterial system.3,4 Following an acute increase in afterload, increased contractility will ensue in order to overcome the rise in afterload and end systolic and diastolic volumes, thus maintaining adequate coupling. However, sustained increase in afterload will lead to uncoupling with further RV dilation, increased wall stress, and decreased stroke volume. With chronic increase in PVR, RV hypertrophy is the main compensatory mechanism to reduce wall tension.
Ventricular Interdependence
The normal pericardium is relatively nondistensable, hence acute dilation of one chamber will be at the expense of the other. With increase in RV overload, the interventricular septum is pushed toward the LV predominantly at end diastole (protosystole in volume overload) and at end systole (protodiastole in pressure and volume overload).
Causes of RV Failure
Causes of RV failure were recently summarized by a working group from the European Society of Cardiology.5 Broadly, causes of RV failure can be grouped into conditions that lead to an increase in RV afterload or a reduction in effective RV function; these are summarized in Table 1.
Table 1: Causes of Acute RV Failure
Increase in RV Afterload |
||
Acute Pulmonary Embolism |
Embolism of thrombus, air, fat, or amniotic fluid into pulmonary vasculature. |
|
Chronic Pulmonary Embolism |
Often unrecognized or misdiagnosed as acute pulmonary embolism. |
|
Acute Respiratory Distress Syndrome |
Hypoxemia and hypercapnia triggering pulmonary vascular vasoconstriction associated with microthrombi and acute muscularization of pulmonary vessels. High airway pressure to maintain gas exchange.6 Specific modes of ventilation such as High Frequency Oscillation.7-9 |
|
Sepsis |
With or without ARDS. |
|
Acute Chest Syndrome |
Due to in situ thrombosis, hypoxemia, vasoconstriction, oxidative stress, and nitric oxide scavenging. |
|
Left-Sided Cardiac Failure |
Increase in left atrial pressure leading to passive increase in pulmonary pressures. May also be associated with an increase in transpulmonary gradient (reflecting an increase in PVR). |
|
Correction of Congenital Pathologies |
Ebstein's anomaly, tetralogy of Fallot, atrial septal defect, or ventricular septal defect closure. |
|
General Surgical Procedures |
||
|
Lobectomy, pneumonectomy |
Loss of effective cross-sectional area of the pulmonary vasculature. |
|
Upper abdominal surgery |
Post-operative atelectasis leading to hypoxic pulmonary vasoconstriction. |
|
Laparoscopic surgery |
Effects of hypercapnia, Trendelenburg position, and positive end-expiratory pressure. |
Reduction in Effective RV Function |
||
Decompensated Pulmonary Hypertension |
Precipitated by infection, anemia, trauma, surgery, pregnancy, arrhythmias, lack of compliance to medication, or maladaptive RV compensation. |
|
RV Infarction |
Coronary occlusion or ischaemia from imbalance between supply and demand (e.g., from increase in RV wall tension and increase RV afterload). |
|
Cardiac Arrest |
Irrespective of the cause of cardiac arrest. |
|
Cardiac Surgery |
||
|
Post Heart Transplant |
The naïve RV may not tolerate pre-existing pulmonary vascular disease. |
|
Tricuspid Valve Surgery |
Replacement or valvuloplasty. |
|
Inadequate myocardial protection during any cardiac intervention. |
|
Diagnosis
Clinical features include signs of RV pressure overload (RV sternal lift, loud pulmonic component of the second heart sound), congestion with elevated jugular venous pressure, peripheral edema, and low cardiac output. Chest X-ray and electrocardiogram may be normal, or the electrocardiogram may show signs of RV strain or ischaemia.
RV function can be readily assessed with echocardiography.11,12 In case of RV failure, severe RV and right atrial dilation are usually present and are associated with decreased markers of systolic function, such as TAPSE (Tricuspid Annular Plane Systolic Excursion), fractional area of change, and myocardial velocities at the tricuspid annulus. Significant tricuspid regurgitation (TR) is usually present due to annular dilatation. Estimation of RV systolic pressure can be obtained from a TR jet; however, it may be invalid in case of severe regurgitation. Presence of estimated RV systolic pressure beyond 60 mmHg suggests chronicity.13 Associated findings include dilated inferior vena cava with minimal respiratory variation, potentially confounded by positive pressure ventilation. RV hypertrophy, dilated pulmonary artery, and intraventricular septal shift are usually seen in chronic pulmonary hypertension. The presence of a moderate to large pericardial effusion is an ominous finding. Newer parameters such as strain and three-dimension may help for more accurate quantification of RV failure.
In the setting of RV myocardial infarction, wall motion abnormalities may be observed. McConnell's sign is a type of wall motion abnormality with hypokinesis of the mid free RV wall and hyperkinesia of the RV apex and annulus. It can also be present with any acute increase in PVR, irrespective of the etiology.14 Other imaging techniques such as chest computed tomography and magnetic resonance imaging may not be feasible in case of severe hemodynamic instability, in which case echocardiography remains the technique of choice.
Low cardiac output measured with a pulmonary artery catheter may be confounded by presence of severe TR. Similarly, low pulmonary arterial pressures may indicate worsening RV failure. Low mixed venous saturation will inform on the inadequacy between oxygen delivery and demand.
Treatment
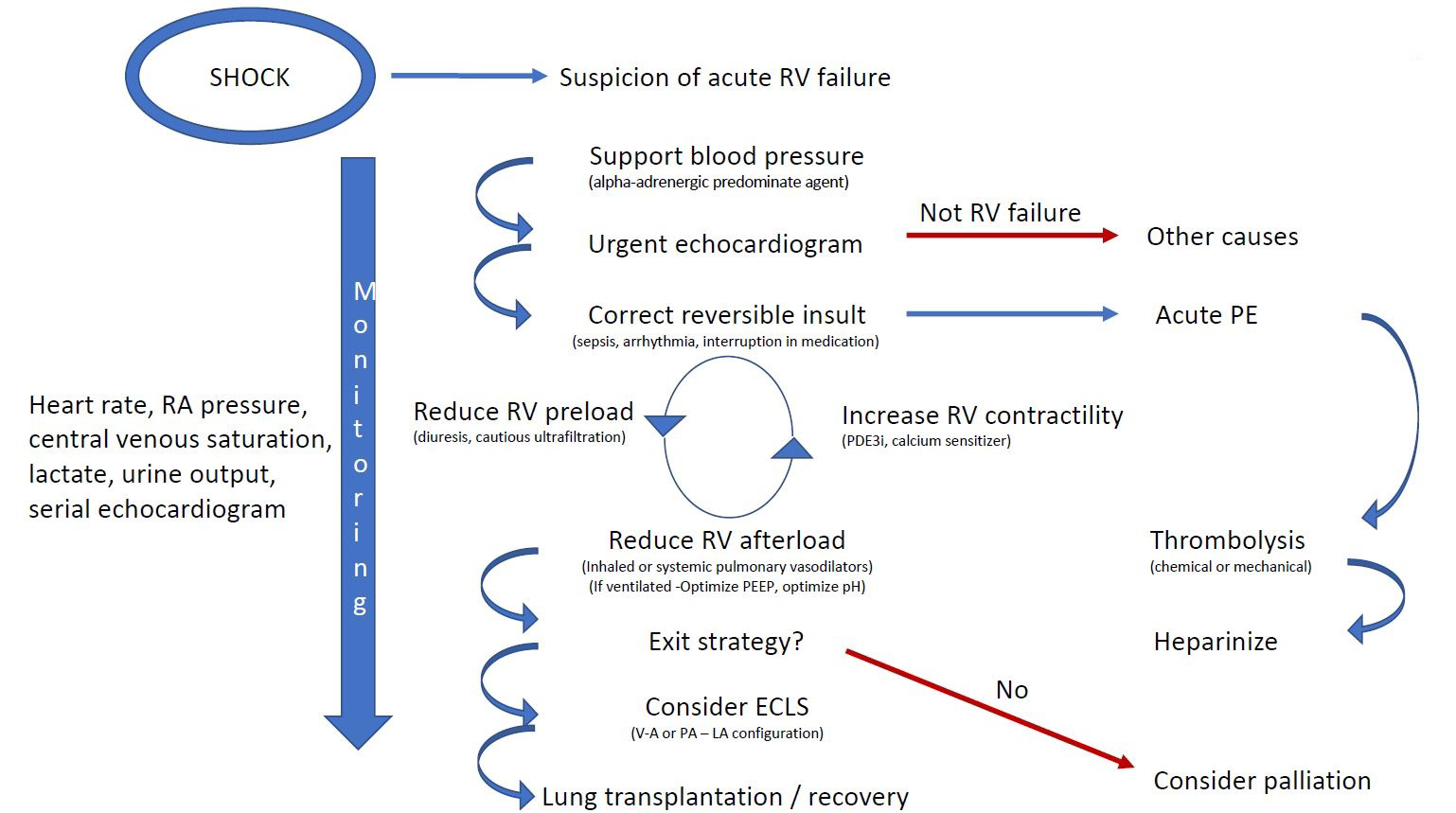
Treatment goals include maintenance of systemic blood pressure, reversal/stabilization of potential exacerbating conditions, and improvement in RV function (Figure 1). A strategy to improve RV function must consider optimizing pre-load, afterload, and contractility.1,15 A successful strategy will lead to improvement in systemic oxygen delivery by increasing forward flow through the pulmonary vasculature and reducing the adverse influence of RV dilation and septal shift on LV function.
Figure 1
Monitoring
It is important to assess the effects of interventions on RV function, volume, and oxygen delivery. Although these patients are unstable and difficult to manage, it is unclear that invasive hemodynamic monitoring provides more information than that obtained from readily available and less-invasive methods. A central venous catheter will facilitate administration of vaso-active medications and allow for estimation of right atrial pressure and central venous saturations. An arterial line will facilitate measurements of systemic blood pressure and obtaining markers of tissue oxygenation (lactate) and arterial blood gasses (pH, PaCO2, and PaO2). Monitoring heart rate and systemic pulse pressure/waveform may allow for inferences regarding changes in stroke volume. Regular assessment of RV size and function and influence of RV on LV filling can be obtained using echocardiography.
Correct Precipitating Causes
Precipitating causes should be reversed or stabilized. In the setting of acute coronary ischaemia, revascularization may restore RV function. Atrial arrhythmias can destabilize patients with chronic RV failure from pulmonary hypertension.16 Failure to restore sinus rhythm is associated with poor prognosis. In shock state, direct current cardioversion is indicated, and medical therapies can be used to either help restore sinus rhythm in non-shock states or reduce the risk of recurrence. The use of agents to control heart rate (e.g., beta-blockers and calcium channel blockers) may, however, have direct negative inotropic effects and may not be tolerated. Correcting hypercapnia and reducing hypoxemia may reduce RV afterload.
Surgery
Surgery and attendant anaesthesia places additional stress on the RV and is beyond the scope of this review but has been addressed by other authors.5 In general, the same aforementioned principles apply in the intra-operative and post-operative period. Familiarization of potential effects of medications on RV and PVR is important.
Preserve Systemic Blood Pressure
RV dilation will lead to a progressive decrease in RV coronary perfusion. The increase in wall tension will also lead to increase in myocardial oxygen consumption. The extent to which the reduction in coronary blood flow by capillary rarefaction and oxygen utilization by abnormal RV metabolism in states of chronic RV overload, which is exacerbated in an acutely decompensated RV, is unknown. Because pulmonary vasodilators have off-target effects on the systemic vasculature and may cause systemic hypotension, it is important to initiate a vasoconstrictor agent prior to using systemically administered pulmonary vasodilators.
In general, alpha-adrenergic agents are recommended. Although these agents may increase pulmonary vascular tone, this seems to be overshadowed by the positive effects on systemic pressure in experimental models of RV failure. Low-dose vasopressin has been advocated because it may restore systemic pressure without increasing the heart rate. However, higher doses may have an adverse effect on coronary and pulmonary blood flow. Agents with predominant beta-adrenergic effects should be used with caution because these may induce tachycardia with subsequent reduction in diastolic filling time.
RV Preload
Optimization of RV preload is important. In acute RV infarction, administration of fluids may be required to increase venous return and maintain pulmonary perfusion. However, overdistention of an ischaemic RV may worsen cardiac function.
Diuresis may be one of the most important treatment strategies in patients with decompensated and dilated RV. A reduction in RV volume should lead to a reduction in RV wall tension, thereby reducing myocardial oxygen consumption and improving coronary blood flow to the RV. In addition, a reduction in RV volume may lead to a reduction in TR. Diuresis will also reduce the influence of the RV on LV diastolic filling by reducing intra-pericardial pressure and reducing septal shift. Improvement in LV filling may lead to improved cardiac function and oxygen delivery and should improve renal perfusion, even in patients with cardio-renal syndrome and acute renal dysfunction.
Reduce RV Afterload
PVR is conventionally considered as representing RV afterload. However, pulmonary vascular compliance and impedance as well as left atrial pressure are other forces that constitute RV afterload.17 Any event that reduces the cross-sectional area of the pulmonary vasculature (pulmonary embolism) or vasoconstriction (hypoxemia and hypercapnia) or that causes an increase in left atrial pressure will induce an increase in RV afterload. RV afterload may also be increased by atelectasis. Improving atelectasis may have a significant benefit to RV function by reducing the effects of hypoxic pulmonary vasoconstriction on RV afterload. Therefore, patients with RV failure from pulmonary vascular disease may benefit from judicious titration of positive end-expiratory pressure. Positive end-expiratory pressure must be applied with caution because alveolar overdistention may also load the RV by compressing or stretching the pulmonary capillaries.6
Pulmonary vasodilators may be beneficial in acute RV failure. The ideal pulmonary vasodilator would have a rapid onset of action and short half-life and be selective to the pulmonary vasculature. In that regard, the use of inhaled pulmonary vasodilators is attractive. Nitric oxide, prostanoids, and PDE5 inhibitors have been reported to improve RV afterload. The use of systemic agents is limited by a longer half-life and off-target effects on the systemic vasculature as well as potentially increased intra-pulmonary shunt by non-selective pulmonary vasodilation.
Our recommendation is to consider using inhaled nitric oxide or PGI2 in conjunction with an oral or intravenous PDE5 inhibitor. For naïve patients with World Health Organization group I pulmonary hypertension, parenteral prostanoids should be considered because they may salvage patients who present de-novo with advanced pulmonary arterial hypertension and RV failure.
Improve Contractility
The agents to increase RV contractility are the same as those used to treat LV failure. Their effectiveness may be limited by two constraints. First, the RV is less muscular than the LV. Second, the ischaemic, overloaded RV may have limited contractile reserve. Though PDE3 inhibitors (e.g., milrinone) and calcium channel sensitizers (e.g., levosimendan) have been shown to improve RV function acutely, there are no large outcome-based studies to support one strategy over another. The use of PDE3 inhibitors may also be limited by systemic vasodilation and hypotension. Epinephrine may also be used; however, it can lead to an undesired tachycardia.
Heart Rate
Tachycardia will lead to an increase in myocardial oxygen consumption and may effectively reduce cardiac output by reducing diastolic filling time. The effects of tachycardia are compounded in the setting of RV overload when RV systolic duration increases and may further overlap and impair diastolic LV filling. There is likely an optimal heart rate for patients with acute RV failure. During resuscitation, we use increases in heart rate as a marker of progressive worsening or as a cautionary sign that our treatments may not be having their desired effect.
Mechanical Circulatory Support
It is important to recognize patients who are failing medical therapy before they develop refractory organ dysfunction. Mechanical circulatory support should be considered in patients who have an exit strategy. This includes lung transplant or, in a treatment-naïve patient with pulmonary arterial hypertension, sufficient time to allow pulmonary vasodilators to work. In the absence of foreseeable exit strategy, palliative approach and comfort measures should be considered.
There is no optimum configuration for mechanical circulatory support. Considerations include expertise of the center, stability of the situation, and anticipated duration of support.18 In our center, a staged approach is often adopted. Initial peripheral venoarterial configuration may be converted to a pulmonary artery to left atrium configuration through a Novalung (Novalung GmbH; Heilbronn, Germany) membrane in patients who are being considered for lung transplantation to facilitate mobility.
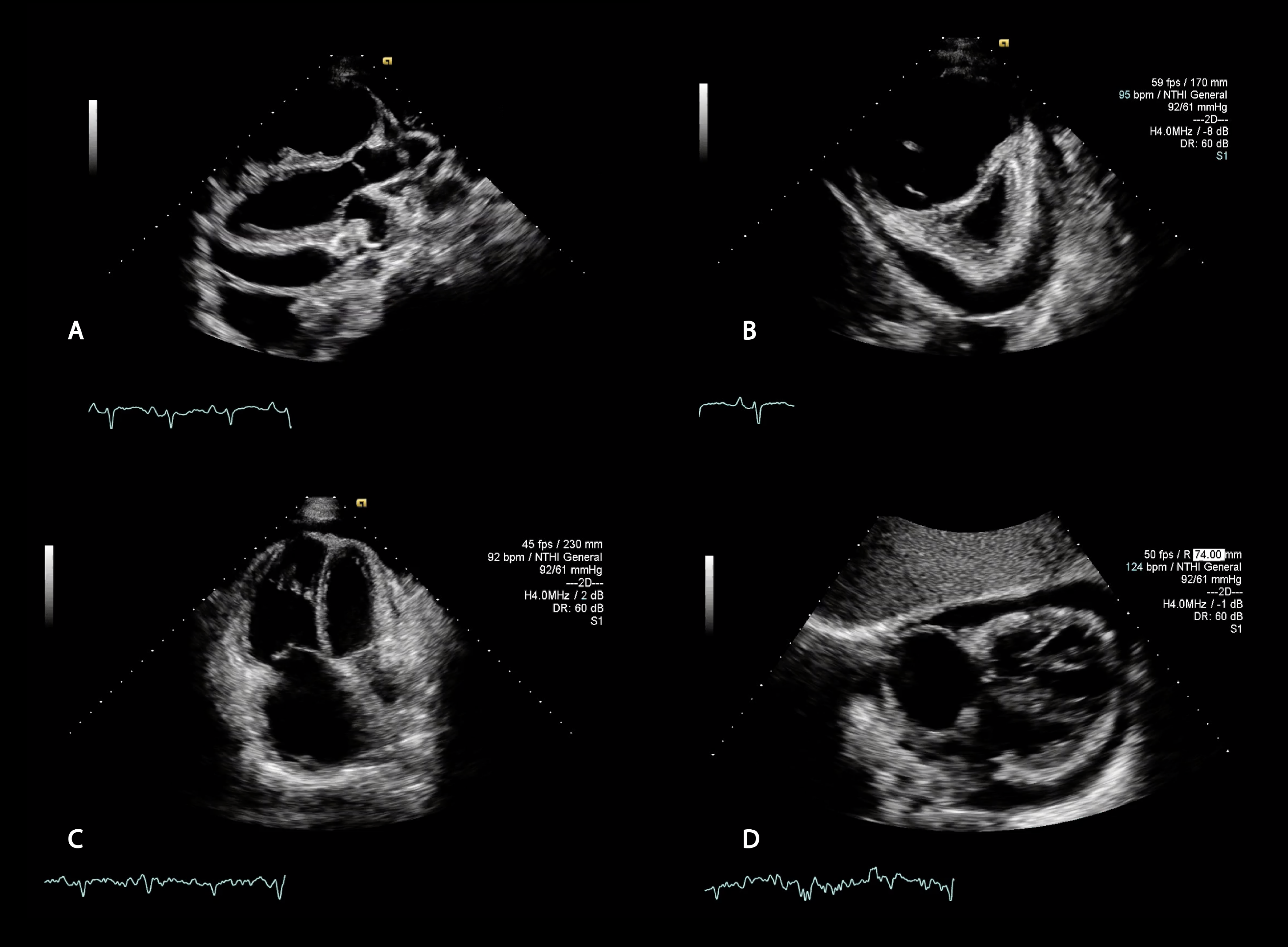
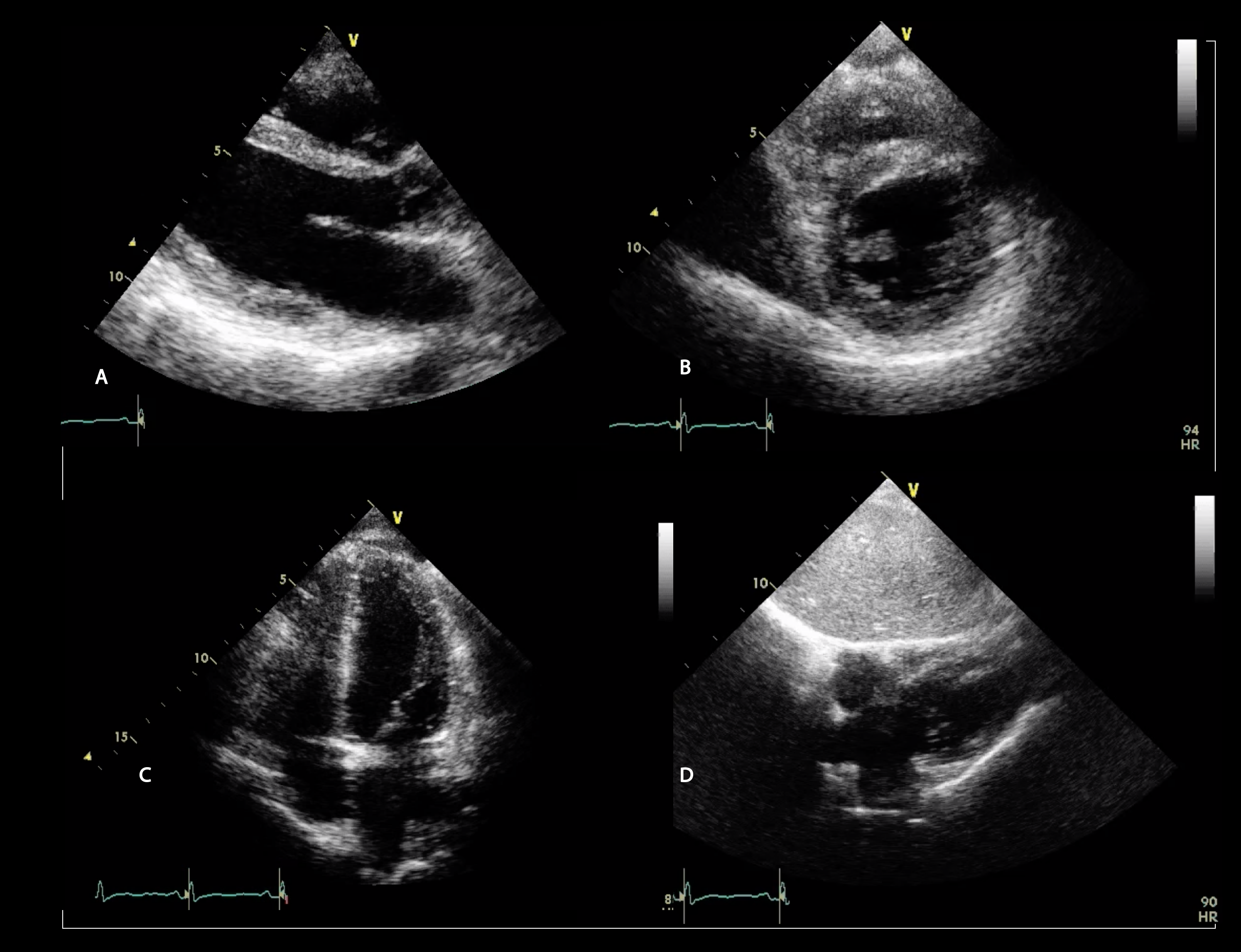
Remarkably, the RV has an incredible capacity to remodel once afterload is improved. Indeed, RV function generally recovers almost fully after lung transplantation (Figures 2-3 and Videos 1-4).19
Figure 2: Transthoracic Views of a Patient With Severe Pulmonary Hypertension
Figure 3: Transthoracic Echocardiography Performed 3 Months After Bilateral Lung Transplant Showing Significant Remodeling of Both Ventricles
Video 1: Parasternal Short-Axis View Before Lung Transplantation
Video 2: Apical Four-Chamber View Before Lung Transplantation
Video 3: Parasternal Short-Axis View After Lung Transplantation
Video 4: Apical Four-Chamber View After Lung Transplantation
References
- Pinsky MR. The right ventricle: interaction with the pulmonary circulation. Crit Care 2016;20:266.
- Haddad F, Couture P, Tousignant C, Denault AY. The right ventricle in cardiac surgery, a perioperative perspective: I. Anatomy, physiology, and assessment. Anesth Analg 2009;108:407-21.
- Vonk Noordegraaf A, Westerhof BE, Westerhof N. The Relationship Between the Right Ventricle and its Load in Pulmonary Hypertension. J Am Coll Cardiol 2017;69:236-43.
- Vieillard-Baron A, Naeije R, Haddad F, et al. Diagnostic workup, etiologies and management of acute right ventricle failure : A state-of-the-art paper. Intensive Care Med 2018;May 9:[Epub ahead of print].
- Harjola VP, Mebazaa A, Čelutkienė J, et al. Contemporary management of acute right ventricular failure: a statement from the Heart Failure Association and the Working Group on Pulmonary Circulation and Right Ventricular Function of the European Society of Cardiology. Eur J Heart Fail 2016;18:226-41.
- Guérin C, Matthay MA. Acute cor pulmonale and the acute respiratory distress syndrome. Intensive Care Med 2016;42:934-6.
- Mekontso Dessap A, Boissier F, Charron C, et al. Acute cor pulmonale during protective ventilation for acute respiratory distress syndrome: prevalence, predictors, and clinical impact. Intensive Care Med 2016;42:862-70.
- Ursulet L, Roussiaux A, Belcour D, et al. Right over left ventricular end-diastolic area relevance to predict hemodynamic intolerance of high-frequency oscillatory ventilation in patients with severe ARDS. Ann Intensive Care 2015;5:25.
- Repessé X, Charron C, Vieillard-Baron A. Acute cor pulmonale in ARDS: rationale for protecting the right ventricle. Chest 2015;147:259-65.
- Kim H, Paek JH, Song JH, et al. Permissive fluid volume in adult patients undergoing extracorporeal membrane oxygenation treatment. Crit Care 2018;22:270.
- Lang RM, Badano LP, Mor-Avi V, et al. Recommendations for cardiac chamber quantification by echocardiography in adults: an update from the American Society of Echocardiography and the European Association of Cardiovascular Imaging. J Am Soc Echocardiogr 2015;28:1-39.e14.
- Rudski LG, Lai WW, Afilalo J, et al. Guidelines for the echocardiographic assessment of the right heart in adults: a report from the American Society of Echocardiography endorsed by the European Association of Echocardiography, a registered branch of the European Society of Cardiology, and the Canadian Society of Echocardiography. J Am Soc Echocardiogr 2010;23:685-713.
- Kurzyna M, Torbicki A, Pruszczyk P, et al. Disturbed right ventricular ejection pattern as a new Doppler echocardiographic sign of acute pulmonary embolism. Am J Cardiol 2002;90:507-11.
- Walsh BM, Moore CL. McConnell's Sign Is Not Specific for Pulmonary Embolism: Case Report and Review of the Literature. J Emerg Med 2015;49:301-4.
- Granton J, Mercier O, De Perrot M. Management of severe pulmonary arterial hypertension. Semin Respir Crit Care Med 2013;34:700-13.
- Olsson KM, Nickel NP, Tongers J, Hoeper MM. Atrial flutter and fibrillation in patients with pulmonary hypertension. Int J Cardiol 2013;167:2300-5.
- Tedford RJ. Determinants of right ventricular afterload (2013 Grover Conference series). Pulm Circ 2014;4:211-9.
- Machuca TN, de Perrot M. Mechanical Support for the Failing Right Ventricle in Patients With Precapillary Pulmonary Hypertension. Circulation 2015;132:526-36.
- Gorter TM, Verschuuren EAM, van Veldhuisen DJ, et al. Right ventricular recovery after bilateral lung transplantation for pulmonary arterial hypertension†. Interact Cardiovasc Thorac Surg 2017;24:890-7.
Clinical Topics: Arrhythmias and Clinical EP, Cardiac Surgery, Congenital Heart Disease and Pediatric Cardiology, Diabetes and Cardiometabolic Disease, Heart Failure and Cardiomyopathies, Invasive Cardiovascular Angiography and Intervention, Noninvasive Imaging, Pericardial Disease, Prevention, Pulmonary Hypertension and Venous Thromboembolism, Implantable Devices, SCD/Ventricular Arrhythmias, Atrial Fibrillation/Supraventricular Arrhythmias, Cardiac Surgery and Arrhythmias, Cardiac Surgery and CHD and Pediatrics, Cardiac Surgery and Heart Failure, Congenital Heart Disease, CHD and Pediatrics and Arrhythmias, CHD and Pediatrics and Imaging, CHD and Pediatrics and Interventions, CHD and Pediatrics and Prevention, CHD and Pediatrics and Quality Improvement, Acute Heart Failure, Heart Transplant, Pulmonary Hypertension, Interventions and Imaging, Interventions and Structural Heart Disease, Interventions and Vascular Medicine, Echocardiography/Ultrasound, Magnetic Resonance Imaging, Hypertension, Stress
Keywords: Hypertension, Pulmonary, Pregnancy, Pulmonary Artery, Heart Septal Defects, Ventricular, Blood Pressure, Acute Chest Syndrome, Respiratory Distress Syndrome, Cardiac Output, Low, Vena Cava, Inferior, Stroke Volume, Nitric Oxide, Tricuspid Valve Insufficiency, Pneumonectomy, Hyperkinesis, Hypercapnia, Pericardial Effusion, Amniotic Fluid, Atrial Fibrillation, Vasoconstriction, Diastole, Head-Down Tilt, Arterial Pressure, Coronary Occlusion, Atrial Pressure, Dilatation, Heart Sounds, Electrocardiography, X-Rays, Heart Failure, Infarction, Tricuspid Valve, Pulmonary Embolism, Vascular Resistance, Positive-Pressure Respiration, Thrombosis, Heart Septal Defects, Atrial, Myocardial Infarction, Hypertrophy, Right Ventricular, Heart Transplantation, Pulmonary Atelectasis, Magnetic Resonance Imaging, Echocardiography, Sepsis, Edema, Oxidative Stress, Prognosis, Tomography, Laparoscopy, Heart Arrest, Pericardium, Oxygen, Systole, Cross-Sectional Studies, Tetralogy of Fallot, Venous Pressure
< Back to Listings