Current Status of the ABI in Diagnosis, Risk Assessment and Screening
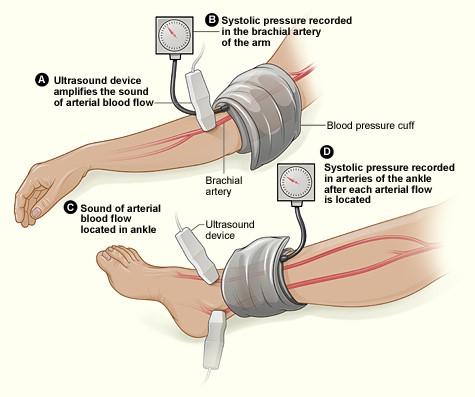
The ankle brachial index (ABI) is a simple tool for identifying lower extremity peripheral artery disease (PAD). The ABI is measured in each leg using a blood pressure cuff placed at the ankle and a hand-held doppler ultrasound device to identify systolic blood pressure at both pedal arteries (dorsalis pedis and posterior tibial arteries). The cuff and doppler device are then used to measure the systolic pressure in both brachial arteries. The ABI is the result of dividing the highest pedal systolic pressure in the foot to the highest systolic pressure in the brachial arteries. PAD is defined as an ankle brachial index < 0.90 (Figure 1).1
Figure 1
Diagnosis
The clinical utility of the ABI depends on the context of its use. The ABI is used as a diagnostic test in an individual with leg symptoms or signs that could be PAD. Symptoms of claudication due to PAD vary widely from the classic muscle pain (e.g. calf pain), to leg weakness, or other leg discomfort, but virtually all claudication symptoms are associated with activity and relieved by rest.2 Critical limb ischemia (chronic rest pain, ischemic ulcers or gangrene) or acute limb ischemia (sudden painful, pale leg progressing to foot or leg paresthesia or paralysis) are suspected on clinical symptoms and signs.3 A diagnosis of PAD will lead to therapies that hopefully improve leg symptoms and function and prevent the progression of disease. Examples include structured exercise programs, Cilostazol, and when appropriate, revascularization. Lowering cardiovascular risk factors is important for preventing PAD progression and has other benefits, including decreasing the risk of cardiac events and stroke.
Risk Assessment
Measuring an ABI for risk assessment refers to its use in patients at elevated cardiovascular risk (e.g. smokers, or patients with coronary disease), but who do not have leg symptoms. In these patients, the diagnosis of asymptomatic PAD elevates their cardiovascular risk further. The utility of measuring ABI in this situation depends on whether an abnormal ABI leads to interventions that would not have been offered and that reduce the risk of myocardial infarction and stroke, such as goal directed medical therapies including statins and antiplatelet therapy.
Screening
Screening refers to the use of ABI in asymptomatic individuals at low or moderate cardiovascular risk and who do not have leg symptoms. The assumption is that finding asymptomatic PAD will lead to interventions to lower cardiovascular risk.
Accuracy of ABI
The accuracy of any diagnostic or screening test depends on the test characteristics (i.e. sensitivity and specificity for identifying PAD), and the prevalence of disease (as this affects positive and negative predictive value). In several studies, the sensitivity of an ABI measured at rest is about 68-84% and the specificity is about 84%-99%.4 Measuring the ABI after exercising (e.g. walking on a treadmill) increases the sensitivity of the test for identifying PAD by about another 25%.4 In US and European guidelines, the resting and exercise ABI have class I indications for assessing PAD in patients with leg symptoms.5,6 A reasonable diagnostic strategy for patients with leg symptoms is to measure the resting ABI, and if normal, measure an exercise ABI.4
In the patient without leg symptoms or signs of PAD, the ABI can identify asymptomatic PAD in patients at high risk of cardiovascular morbidity and mortality. However, in the US guidelines, measuring ABI in asymptomatic patients is a class IIa recommendation (reasonable).6 The IIa recommendation is based on the fact that these patients warrant intensive atherosclerosis risk factor reduction regardless of the ABI. Moreover, as the prevalence of PAD is lower in asymptomatic high cardiovascular risk patients than patients with leg symptoms, the false positive rate will be higher and could lead to unnecessary further testing (e.g. angiography by computed tomography, magnetic resonance, or invasive means). The European guidelines do not even comment on the use of ABI in this setting.5
The prevalence of PAD is even lower in patients who are not at high cardiovascular risk, e.g. screening for PAD in the general population, and the false positive rate for ABI is fairly high. The US guidelines give ABI screening in patients without leg symptoms or high cardiovascular risk a class III recommendation (no benefit or possible harm),6 and is not recommended by the US Preventive Services Task Force.7
ABI Screening to Change Cardiovascular Risk
The questionable value of using ABI in patients without leg symptoms or signs seems a bit odd to some pundits.8 Older trials looking at the value of screening asymptomatic people with the ABI and treating PAD patients with aspirin found no benefit on cardiovascular morbidity or mortality.9,10 One recent study that offered bundled screening for abdominal aortic aneurysms, PAD, and hypertension in Danish men 65-74 years of age did find that the screened group were more likely to be taking risk reducing therapies at 6 months and had a lower death rate over 4.4 years.11 However, it is uncertain which screening test was responsible for this benefit.12 Although some argue that knowledge of asymptomatic PAD is a motivator for patients to take antihypertensive or lipid lowering treatment, this is speculative. An alternative explanation is that patients take their antihypertensive medications because they are told their blood pressure is high rather than their ABI being low.
PAD clusters with smoking, high cholesterol, hypertension, and diabetes and patients with these conditions warrant intensive risk factor modification. Therefore, the incremental value of ABI screening in the general population is to identify the very few people with asymptomatic PAD who don't have cardiovascular disease or risk factors. So, the incremental yield of ABI screening is marginal and health resources are probably better directed at other actions to promote health.
Overall, the US and European guidelines are very similar and support the ABI as a diagnostic tool in patients with leg symptoms, but they are less enthusiastic about its use to discover PAD in patients at elevated cardiovascular risk without leg symptoms.5,6 Screening for PAD in a low to moderate risk asymptomatic population is not supported by either guideline.
References
- Aboyans V, Criqui MH, Abraham P, et al. Measurement and interpretation of the ankle-brachial index: a scientific statement from the American Heart Association. Circulation 2012;126:2890-09.
- McDermott MM, Greenland P, Liu K, et al. Leg symptoms in peripheral arterial disease: associated clinical characteristics and functional impairment. JAMA 2001;286:1599-606.
- Thukkani AK, Kinlay S. Endovascular intervention for peripheral artery disease. Circ Res 2015;116:1599-613.
- Aday AW, Kinlay S, Gerhard-Herman MD. Comparison of different exercise ankle pressure indices in the diagnosis of peripheral artery disease. Vasc Med 2018:1358863X18781723 [Epub ahead of print].
- Aboyans V, Ricco JB, Bartelink MEL, et al. 2017 ESC Guidelines on the Diagnosis and Treatment of Peripheral Arterial Diseases, in collaboration with the European Society for Vascular Surgery (ESVS): Document covering atherosclerotic disease of extracranial carotid and vertebral, mesenteric, renal, upper and lower extremity arteries Endorsed by: the European Stroke Organization (ESO)The Task Force for the Diagnosis and Treatment of Peripheral Arterial Diseases of the European Society of Cardiology (ESC) and of the European Society for Vascular Surgery (ESVS). Eur Heart J 2018;39:763-816.
- Gerhard-Herman MD, Gornik HL, Barrett C, et al. 2016 AHA/ACC guideline on the management of patients with lower extremity peripheral artery disease: Executive Summary: a report of the American College of Cardiology/American Heart Association Task Force on Clinical Practice Guidelines. J Am Coll Cardiol 2017;69:1465-1508.
- US Preventive Services Task Force, Curry SJ, Krist AH, et al. Risk assessment for cardiovascular disease with nontraditional risk factors: US Preventive Services Task Force Recommendation Statement. JAMA 2018;320:272-80.
- Beckman JA. The U.S. Preventive Services Task Force: objectivity as authority. J Am Coll Cardiol 2019;73:1719-22.
- Belch J, MacCuish A, Campbell I, et al. The prevention of progression of arterial disease and diabetes (POPADAD) trial: factorial randomised placebo controlled trial of aspirin and antioxidants in patients with diabetes and asymptomatic peripheral arterial disease. BMJ 2008;337:a1840.
- Fowkes FG, Price JF, Stewart MC, et al. Aspirin for prevention of cardiovascular events in a general population screened for a low ankle brachial index: a randomized controlled trial. JAMA 2010;303:841-48.
- Lindholt JS, Sogaard R. Population screening and intervention for vascular disease in Danish men (VIVA): a randomised controlled trial. Lancet 2017;390:2256-65.
- Ayoub C, Murad MH. Population-based screening for vascular disease. Lancet 2017;390:2218-20.
Clinical Topics: Cardiovascular Care Team, Dyslipidemia, Invasive Cardiovascular Angiography and Intervention, Noninvasive Imaging, Prevention, Vascular Medicine, Atherosclerotic Disease (CAD/PAD), Lipid Metabolism, Nonstatins, Novel Agents, Statins, Interventions and Imaging, Interventions and Vascular Medicine, Angiography, Echocardiography/Ultrasound, Nuclear Imaging, Exercise, Hypertension
Keywords: Blood Pressure, Ankle Brachial Index, Risk Factors, Peripheral Arterial Disease, Antihypertensive Agents, Hydroxymethylglutaryl-CoA Reductase Inhibitors, Brachial Artery, Aspirin, Myalgia, Gangrene, Tibial Arteries, Cardiovascular Diseases, Aortic Aneurysm, Abdominal, Paresthesia, Ulcer, Health Resources, Diagnostic Tests, Routine, Blood Pressure Determination, Hypertension, Myocardial Infarction, Stroke, Risk Assessment, Diabetes Mellitus, Angiography, Coronary Disease, National Institutes of Health (U.S.), Ultrasonography, Doppler, Magnetic Resonance Spectroscopy, Lipids, Exercise Therapy, Cholesterol, Tomography
< Back to Listings