Spontaneous Coronary Artery Dissection: Clinical Considerations in Diagnosis and Treatment
Introduction
Spontaneous coronary artery dissection (SCAD) is a unique clinical entity and an important cause of myocardial infarction (MI), particularly among young and middle-aged women. SCAD is defined as a non-iatrogenic, non-traumatic separation of the coronary artery wall, not associated with atherosclerosis.1 The predominant mechanism of myocardial injury is coronary artery obstruction caused by an intramural hematoma (IMH) or intimal disruption compromising the true lumen at the site of dissection.2 The original studies implicated SCAD as the cause of acute coronary syndrome in 0.1% to 4% of cases, but more recent studies have shown SCAD to be the underlying cause of MI in 22-43% of women <50 years.3,4 Although remarkable progress has been made in our understanding of SCAD in recent years, a significant proportion of patients continue to be misdiagnosed and improperly treated. Here we summarize the unique considerations in the diagnosis and management of SCAD with a focus on interventional therapy.
Diagnosis
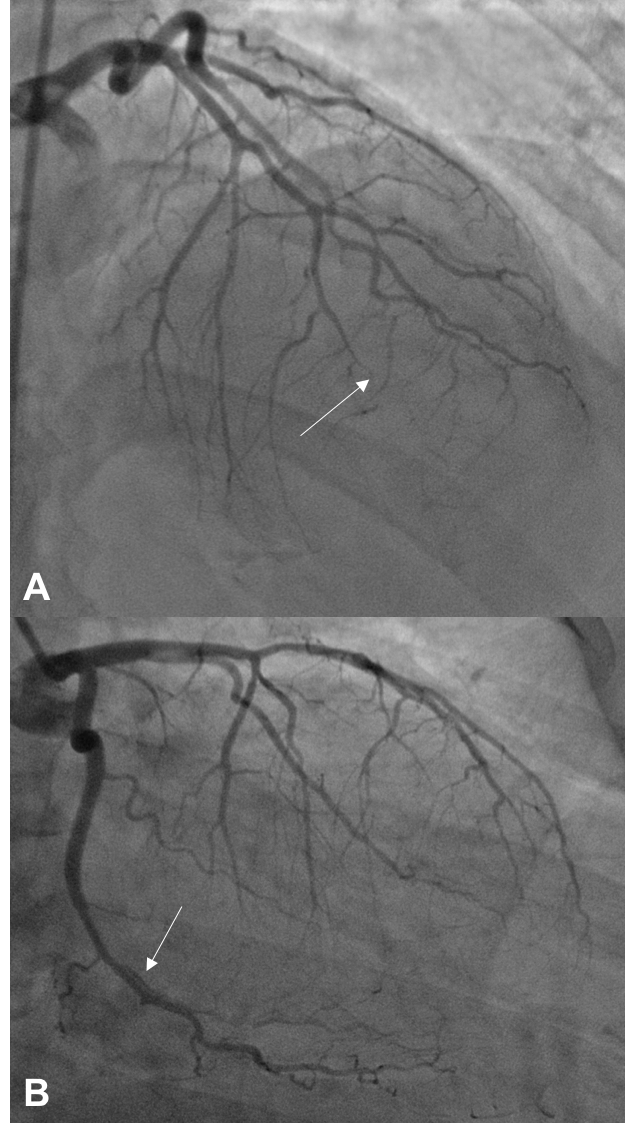
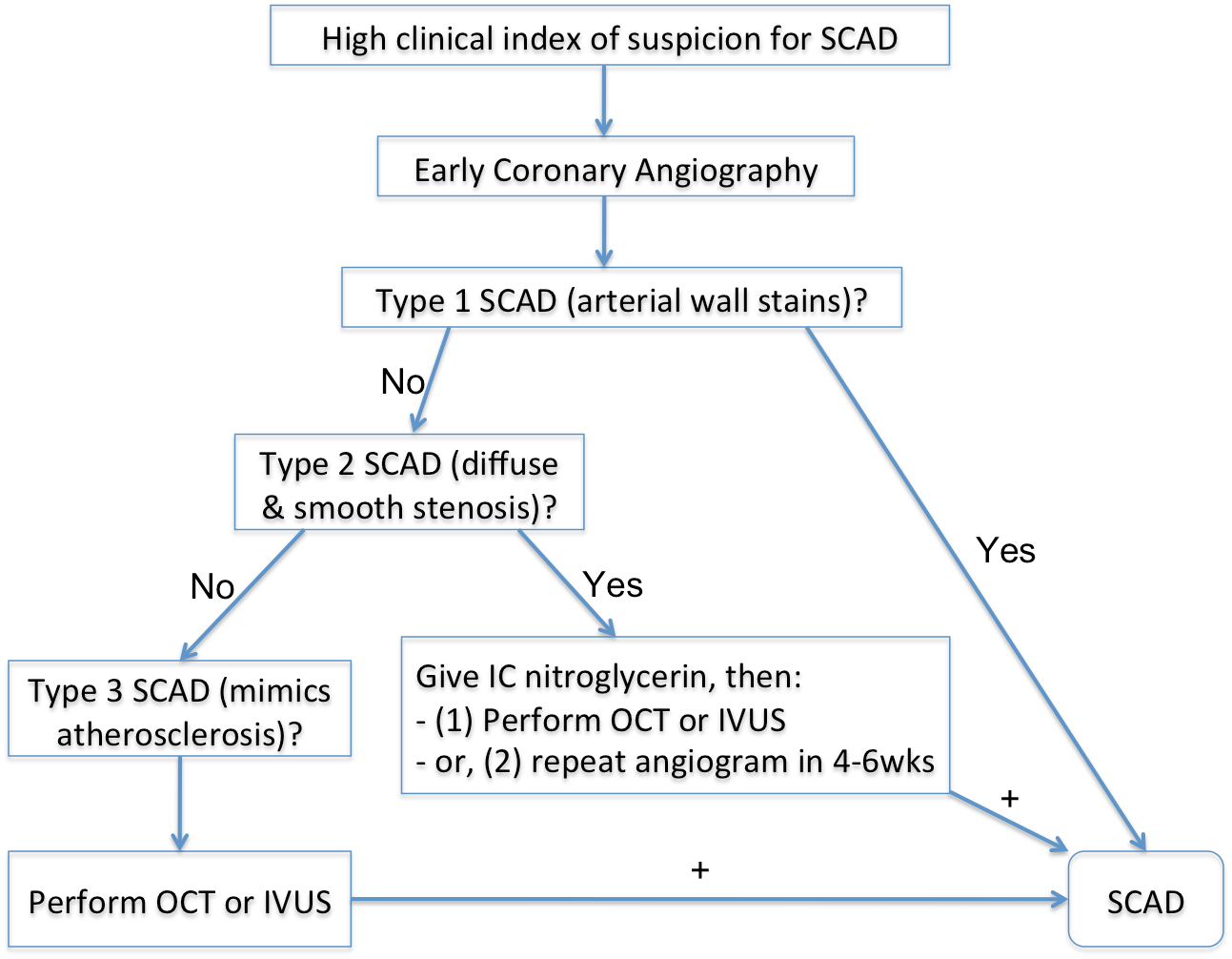
Early and accurate diagnosis of SCAD is vital because the management strategy employed in this condition significantly differs from that of atherosclerotic disease.1 Because patients with SCAD are generally younger and possess less-conventional cardiovascular risk factors, they are often more prone to misdiagnosis. Coronary angiography is the first-line diagnostic tool for patients presenting with acute coronary syndrome due to suspected SCAD and should be performed as early as possible. Pathognomonic angiographic features of SCAD may include multiple radiolucent lines, contrast staining, false lumen appearance, and late contrast clearing, all of which are indicative of intimal tear (type 1 angiographic SCAD) (Figure 1). Intracoronary imaging methods such as optical coherence tomography or intravascular ultrasound can serve as an important tool in increasing the diagnostic yield when there is uncertainty on coronary angiography, especially for cases due to IMH without intimal tear (types 2 [long diffuse narrowing] or 3 [mimics atherosclerosis] SCAD). Appropriate patient selection is important when considering the use of intracoronary imaging in suspected SCAD patients given the small but real risk of propagating the dissection or causing guide-catheter-induced dissection. Moreover, utmost care and meticulous techniques should be used once the decision is made to perform. A proposed algorithm for diagnosis of SCAD is shown in Figure 2.
Figure 1
Figure 2: Proposed Algorithm for Diagnosis of SCAD
Treatment
A paucity of data exists regarding optimal management of SCAD due to lack of randomized trials comparing medical therapy and revascularization strategies. Observational data have shown that when repeat angiography is performed, 70-97% of conservatively managed patients displayed angiographic healing of SCAD lesions.5,6 As such, a conservative treatment strategy is recommended in most cases, which includes an extended inpatient monitoring period of 3-5 days. The backbone of long-term medical therapy in SCAD is a combination of aspirin and beta-blocker. Beta-blocker use has been associated with a lower risk of recurrent SCAD (hazard ratio 0.36) and is therefore an essential part of long-term therapy at our institution.1 A detailed discussion of medical therapy in SCAD is available in a recently published review.7
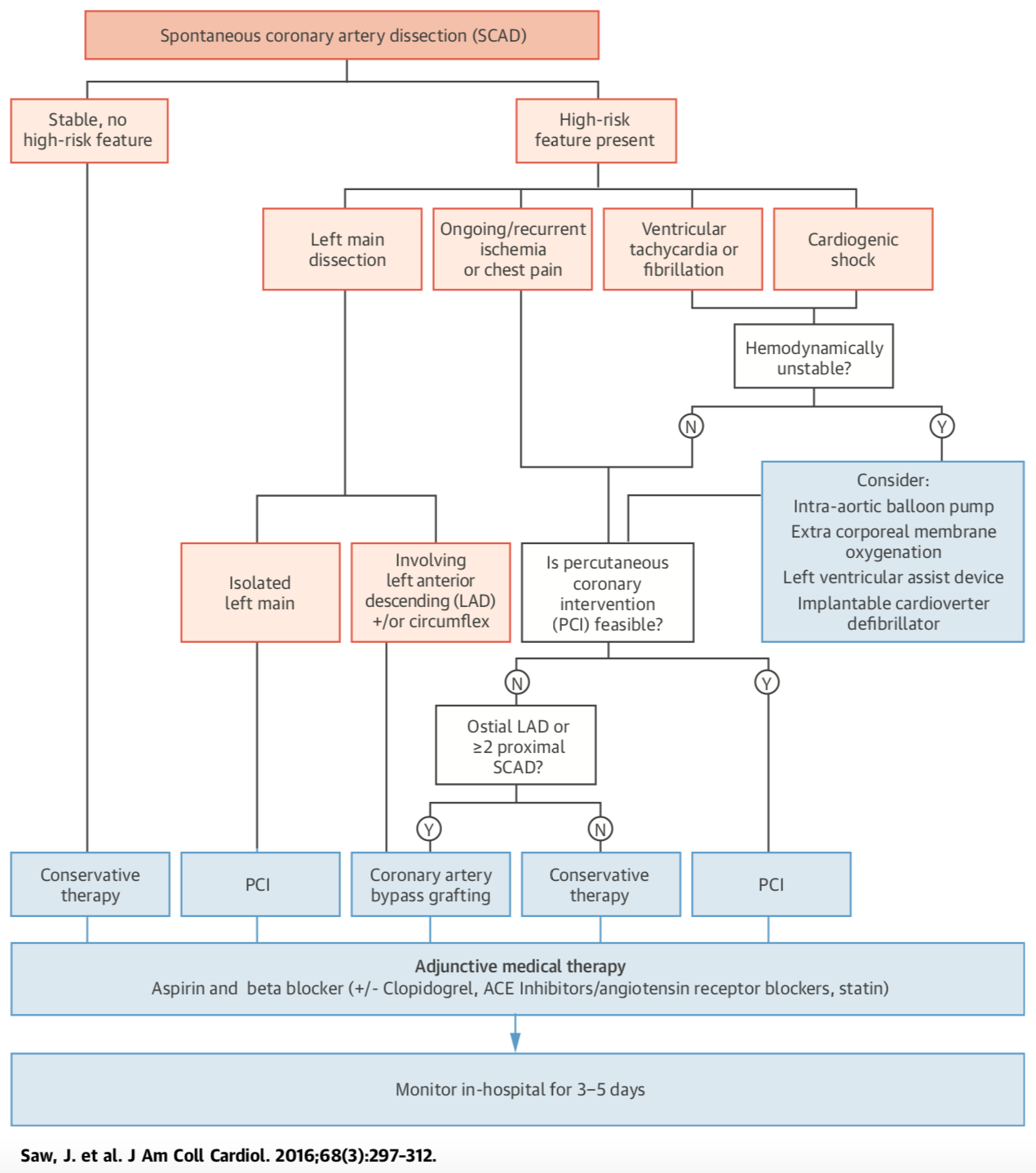
Revascularization for SCAD should be considered in patients exhibiting active myocardial ischemia, hemodynamic instability, or left main dissection, in whom conservative management is an unsatisfactory alternative. Coronary artery bypass grafting surgery (CABG) should be reserved for patients with left main or multivessel proximal large-vessel dissection, especially in the setting of hemodynamic compromise. A proposed algorithm for management of SCAD is shown in Figure 3.
Figure 3: Proposed Algorithm for Management of SCAD Patients
Percutaneous Coronary Intervention in SCAD
According to observational studies, percutaneous coronary intervention (PCI) for treatment of SCAD is associated with lower success rates and higher rates of complications compared with PCI for atherosclerotic CAD.4,5,8,9 In SCAD, affected coronary arteries are more susceptible to iatrogenic dissections. Balloon dilatations or stent placement during PCI can lead to extension of dissections and propagation of IMH, potentially worsening vessel obstruction. Placement of coronary guidewires in the false lumen can lead to the disastrous placement of a stent in the false lumen. Because malapposed stent struts is the most frequently reported cause of stent thrombosis post-PCI,10 the eventual resorption of IMH in SCAD overtime may augment the risk of stent thrombosis. Moreover, due to extensive length of dissections often seen in SCAD, complete revascularization may require long stents, increasing the risk of subsequent in-stent restenosis. A recent retrospective study comparing revascularization strategies and outcomes between patients with ST-segment elevation MI due to SCAD and those with atherosclerotic ST-segment elevation MI reported a mean stent length of 62 ± 37 mm used in SCAD, with 46.78% requiring use of ≥3 stents.9
CanSCAD (the Canadian SCAD Cohort Study), the largest multicenter prospective observational SCAD study, enrolled 750 patients with SCAD from 22 North American centers from 2014 to 2018. The mean length of dissection was 33.2 mm, and the medial stenosis severity was 79.0%. In this study, the majority of patients (86.4%) was managed conservatively initially. Of those, only 13 (2.0%) required subsequent PCI, and 2 (0.3%) required CABG. For those who underwent revascularization as initial therapy, the main reasons for revascularization were ongoing ischemic symptoms (39.1%), ongoing ischemic changes on electrocardiogram (34.5%), and severe luminal stenosis (31.8%). Overall, 110 patients (14.7%) underwent revascularization (14.1% PCI; 0.7% CABG).11 For those who underwent PCI, the technical success rate was relatively low, with 30.1% being unsuccessful and 40.8% achieving partial success. Furthermore, PCI resulted in propagation of SCAD in 32% of patients and improved Thrombolysis in Myocardial Infarction flow in only 57.6% of patients.11
There are several proposed strategies aimed at improving the success rate of PCI in SCAD. For diffuse long lesions, a multi-stent approach may be used to first seal the distal and proximal ends of dissections before stenting the middle segment in order to prevent and minimize IMH propagation.12 Another strategy to avoid antegrade or retrograde prorogation of the IMH is to implant long drug-eluting stents covering in excess of 5-10 mm on both proximal and distal edges of the IMH. Intravascular imaging can provide unique morphological and anatomical details that can be used to avoid certain technical difficulties. For example, optical coherence tomography can confirm positioning of the angioplasty wire in the true lumen and can accurately localize the intimal disruption site and full extent of IMH, thereby facilitating correct stent sizing and deployment. Minimizing balloon inflations or direct stents may also decrease the risk of IMH extension. Moreover, balloon angioplasty alone may also be used to restore coronary flow. A cutting balloon strategy can also be used in an attempt to decompress the blood in the false lumen into the true lumen. Cutting balloon angioplasty should be performed cautiously using an undersized balloon. There are limited data on the effectiveness of these PCI techniques in SCAD.
Conclusion
SCAD is an enigmatic disease, and our understanding of the disease has progressed in recent years, largely owing to several registries including CanSCAD. Although overall survival is excellent after a SCAD event, cardiovascular events do occur following index presentation. Additional studies are required to refine and unify our treatment approaches to patients with SCAD. Advances in diagnostic and therapeutic PCI techniques in the near future may also prove beneficial in treatment of patients with SCAD.
References
- Saw J, Mancini GBJ, Humphries KH. Contemporary Review on Spontaneous Coronary Artery Dissection. J Am Coll Cardiol 2016;68:297-312.
- Hayes SN, Kim ESH, Saw J, et al. Spontaneous Coronary Artery Dissection: Current State of the Science: A Scientific Statement From the American Heart Association. Circulation 2018;137:e523-e557.
- Nishiguchi T, Tanaka A, Ozaki Y, et al. Prevalence of spontaneous coronary artery dissection in patients with acute coronary syndrome. Eur Heart J Acute Cardiovasc Care 2016;5:263-70.
- Nakashima T, Noguchi T, Haruta S, et al. Prognostic impact of spontaneous coronary artery dissection in young female patients with acute myocardial infarction: A report from the Angina Pectoris-Myocardial Infarction Multicenter Investigators in Japan. Int J Cardiol 2016;207:341-8.
- Rogowski S, Maeder MT, Weilenmann D, et al. Spontaneous Coronary Artery Dissection: Angiographic Follow-Up and Long-Term Clinical Outcome in a Predominantly Medically Treated Population. Catheter Cardiovasc Interv 2017;89:59-68.
- Rashid HN, Wong DT, Wijesekera H, et al. Incidence and characterisation of spontaneous coronary artery dissection as a cause of acute coronary syndrome--A single-centre Australian experience. Int J Cardiol 2016;202:336-8.
- Gilhofer TS, Saw J. Spontaneous coronary artery dissection: a review of complications and management strategies. Expert Rev Cardiovasc Ther 2019;17:275-91.
- Lettieri C, Zavalloni D, Rossini R, et al. Management and Long-Term Prognosis of Spontaneous Coronary Artery Dissection. Am J Cardiol 2015;116:66-73.
- Lobo AS, Cantu SM, Sharkey SW, et al. Revascularization in Patients With Spontaneous Coronary Artery Dissection and ST-Segment Elevation Myocardial Infarction. J Am Coll Cardiol 2019;74:1290-300.
- Mori H, Joner M, Finn AV, Virmani R. Malapposition: is it a major cause of stent thrombosis? Eur Heart J 2016;37:1217-9.
- Saw J, Starovoytov A, Humphries K, et al. Canadian spontaneous coronary artery dissection cohort study: in-hospital and 30-day outcomes. Eur Heart J 2019;40:1188-97.
- Saw J, Humphries K, Aymong E, et al. Spontaneous Coronary Artery Dissection: Clinical Outcomes and Risk of Recurrence. J Am Coll Cardiol 2017;70:1148-58.
- Saw J. Coronary angiogram classification of spontaneous coronary artery dissection. Catheter Cardiovasc Interv 2014;84:1115-22.
Clinical Topics: Acute Coronary Syndromes, Cardiac Surgery, Congenital Heart Disease and Pediatric Cardiology, Invasive Cardiovascular Angiography and Intervention, Noninvasive Imaging, Stable Ischemic Heart Disease, Atherosclerotic Disease (CAD/PAD), Aortic Surgery, Cardiac Surgery and CHD and Pediatrics, Cardiac Surgery and SIHD, Congenital Heart Disease, CHD and Pediatrics and Imaging, CHD and Pediatrics and Interventions, CHD and Pediatrics and Quality Improvement, Interventions and ACS, Interventions and Coronary Artery Disease, Interventions and Imaging, Interventions and Structural Heart Disease, Angiography, Echocardiography/Ultrasound, Nuclear Imaging, Chronic Angina
Keywords: Angiography, Coronary Angiography, Percutaneous Coronary Intervention, Acute Coronary Syndrome, Drug-Eluting Stents, Tomography, Optical Coherence, Aspirin, Retrospective Studies, Constriction, Pathologic, Patient Selection, Inpatients, Prospective Studies, Cardiovascular Diseases, Coronary Restenosis, Dilatation, Risk Factors, Coronary Artery Disease, Coronary Vessel Anomalies, Coronary Artery Bypass, Myocardial Infarction, Electrocardiography, Angioplasty, Registries, Atherosclerosis, Angioplasty, Balloon, Hemodynamics, Thrombosis, Algorithms, Diagnostic Errors, Hematoma, Ultrasonography, Interventional, Staining and Labeling, Thrombolytic Therapy, Iatrogenic Disease
< Back to Listings