Transcatheter Aortic Valve Implantation: Durability and Management of Valve Failure
Quick Takes
- Transcatheter aortic valve implantation (TAVI) shows strong midterm durability with low structural valve deterioration rates.
- Valve failure is multifactorial and requires structured imaging surveillance.
- Redo-TAVI is effective in select patients whereas surgery remains necessary for complex failure—underscoring lifetime Heart Team planning.
Transcatheter aortic valve implantation (TAVI) has become a cornerstone therapy for patients with severe aortic stenosis across a wide spectrum of clinical presentations and surgical risk. Initially reserved for patients at prohibitive or high surgical risk, TAVI is now widely adopted in populations at intermediate and low risk following randomized controlled trial (RCT) data demonstrating noninferior or superior outcomes compared with surgical aortic valve replacement (SAVR). As indications progressively expand to younger patients, with more extended anticipated lifespan, assessment of long-term valve durability and the development of strategies for managing transcatheter valve failure have become more critical.
The Valve Academic Research Consortium 3 (VARC-3) consensus was developed as a standardized framework for assessing bioprosthetic valve performance and overcoming the risk of underestimation of previous definitions, based only on the occurrence of major clinical events or reintervention. Bioprosthetic valve dysfunction (BVD) encompasses structural valve deterioration (SVD), nonstructural valve dysfunction, valve thrombosis, and infective endocarditis, with severity stages linked to clinical outcomes and reintervention requirements.1 SVD refers to intrinsic, irreversible changes in the valve prosthesis, including leaflet calcification, fibrosis, or tearing, leading to stenosis or regurgitation. Nonstructural valve dysfunction includes extrinsic causes such as paravalvular leak (PVL), prosthesis-patient mismatch (PPM), or malposition. Bioprosthetic valve failure (BVF) represents clinically significant dysfunction resulting in valve-related death or the need for repeat intervention.1
The establishment of the VARC-3 definition in 2021 enabled consistent reporting of durability outcomes and more meaningful comparisons between TAVI and SAVR.2
Accumulating evidence supports excellent midterm durability of contemporary transcatheter heart valves. Data from registries and RCTs are now available over 5 years of follow-up, showing that >90% of patients remain free from SVD, with severe valve degeneration occurring in <3.5% of patients, of whom only 0.5-1% undergo reintervention.3
Direct comparisons between TAVI and SAVR continue to mature. The NOTION (Nordic Aortic Valve Intervention) trial results demonstrated no difference in the composite endpoint of all-cause death, stroke, or myocardial infarction with TAVI compared with SAVR at 10 years, with no significant difference in BVF, whereas the rate of severe SVD was lower with transcatheter valves (1.5% vs. 10%; hazard ratio 0.2; 95% confidence interval, 0.04-0.7; p= 0.02).4 Additional post hoc analyses suggest lower SVD rates with self-expanding transcatheter valves compared with surgical bioprostheses in select cohorts at intermediate risk.5 Although these findings are encouraging, particularly as TAVI use expands into younger patients, long-term durability beyond 10 years remains unknown, and continued follow-up is essential.
Considerable efforts have focused on identifying the predictors of SVD. Both prosthesis- and patient-related factors interact to influence SVD, pointing to the importance of a tailored approach in the selection of the strategy and device for valve replacement (Figure 1).
Figure 1: Tailored Approach to Selecting the Strategy and Device for Valve Replacement
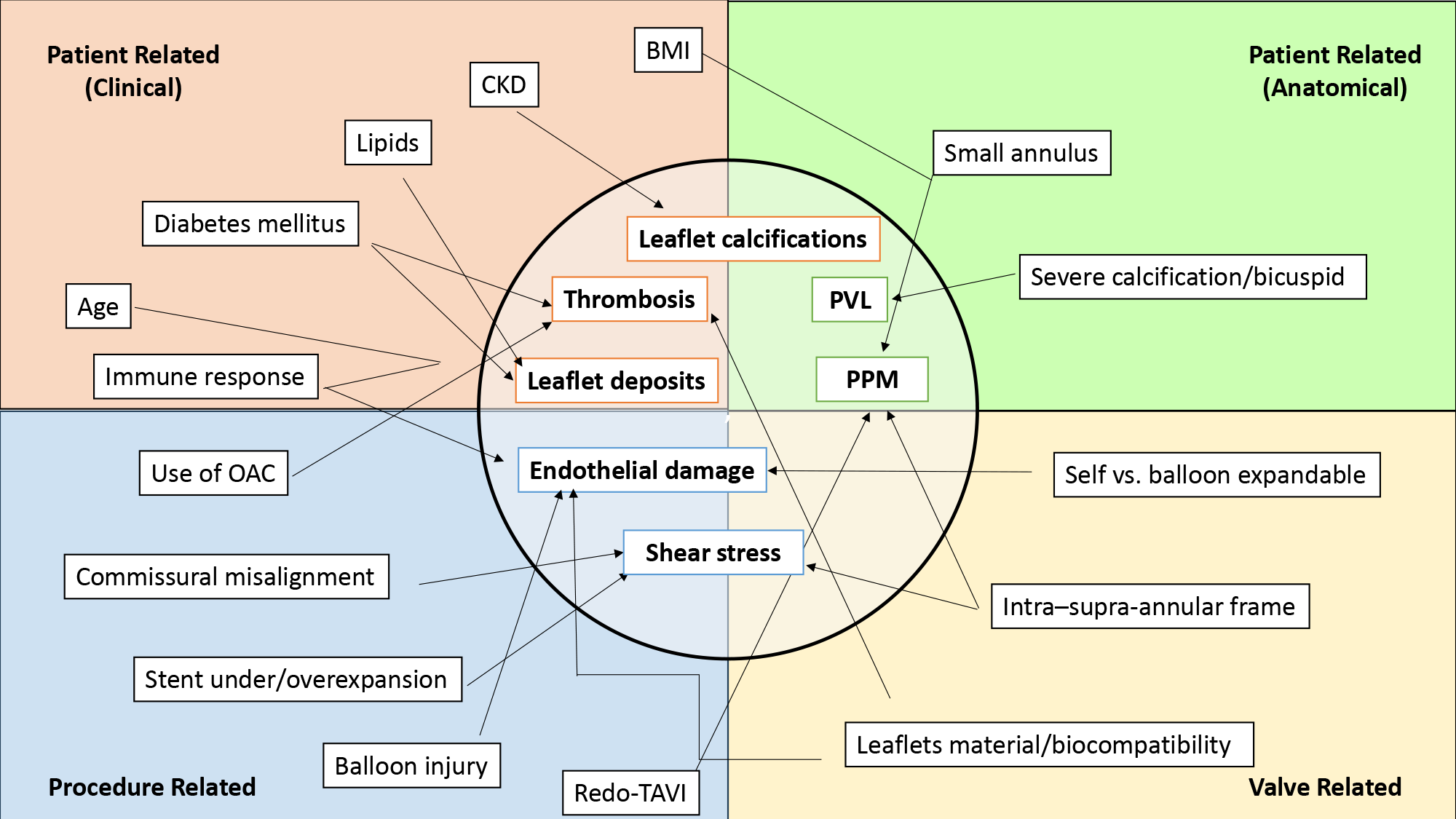
BMI = body mass index; CKD = chronic kidney disease; PVL = paravalvular leak; PPM = prosthesis-patient mismatch; OAC = oral anticoagulant; TAVI = transcatheter aortic valve implantation.
In particular, bicuspid valve anatomy and small annuli are associated with lower procedural success, irrespective of age, increasing the risk of PVL or PPM and still representing the most challenging subsets for TAVI. In such scenarios, supra-annular valve implantation may provide superior hemodynamic performance compared with intra-annular positioning and should, therefore, be considered during procedural planning.3,6
Clinical factors (e.g., inflammation and dysmetabolism) promoting oxidative stress, lipid and glycated products infiltration, and calcium deposition, in addition to prothrombotic status, have been reported to accelerate SVD.3 No pharmacological measure has been approved to date to prevent BVD; regarding the progression of aortic stenosis itself, however, the establishment of individualized antithrombotic strategies, achieving an adequate metabolic control and lowering inflammation, have been suggested to prologue bioprosthetic valve durability.3
Modeling studies suggest that transcatheter valves and SAVR could warrant similar life expectancies in older patients if providing a durability of ≥30% that of surgical valves; in younger patients (40-60 years of age), TAVI valve durability must approach that of surgical bioprostheses to maintain long-term survival benefits.7 As such, durability considerations are critical in a lifetime perspective.
SVD is associated with higher mortality and increased heart failure hospitalizations, highlighting the need for routine follow-up with dedicated imaging.1,2 Transthoracic echocardiography remains the cornerstone of post-TAVI surveillance because SVD definitions require the comparison with early postimplantation findings. Rising transvalvular gradients, new regurgitation, or changes in valve morphology and hemodynamics should prompt further assessment using a multimodality approach. Cardiac computed tomography is typically the next step, helping differentiate structural from nonstructural valve failure and detect leaflet calcification, thrombosis, structural abnormalities, or endocarditis while also providing anatomical details for reintervention planning. Transesophageal echocardiography, cardiac magnetic resonance, and positron emission tomography can offer complementary information to clarify the etiology and better characterize BVD.
Redo-TAVI, or TAVI-in-TAVI, has become a valuable option for select patients with TAVI failure. Registry data show that redo-TAVI is feasible and yields favorable short-term outcomes when patients are carefully selected.8 Preprocedural planning with multimodality imaging is essential to evaluate coronary anatomy, valve geometry, and risks of coronary obstruction or severe PPM. Coronary ostial height, sinus of Valsalva dimensions, and the frame design of the initial valve are key determinants of procedural success.
In cases in which redo-TAVI is contraindicated, including active endocarditis, severe PPM, unfavorable coronary anatomy, or the need for additional cardiac surgery, surgical explantation followed by SAVR may be necessary. These procedures are technically complex and carry substantial operative risk and enhanced 30-day (13.6% vs. 3.4%; p < 0.001) and 1-year mortality (32.4% vs. 15.4%; p = 0.001) compared with less-invasive strategies.9 Although valve-in-valve TAVI is associated with lower early morbidity than surgical explantation, its advantages may diminish over time, with possible increases in late mortality and heart failure hospitalizations beyond 2 years.9 Therefore, referral to experienced centers and multidisciplinary Heart Team evaluation is mandatory for these complex cases.
As TAVI moves into patients who are younger and at lower risk, lifetime valve management should begin at the index procedure, including valve choice, implantation depth, and commissural alignment to facilitate future coronary access and reintervention.9 Scheduled echocardiography is essential for early detection of SVD and, if BVF occurs, integrating clinical, anatomical, and procedural risk should drive the choice between redo-TAVI and surgery. Ongoing registries and RCTs, including trials such as REVALVE (REdo Transcatheter Aortic VALVE Implantation for the Management of Transcatheter Aortic Valve Failure), will further refine optimal management strategies for TAVI failure.10
References
- Patel SP, Garcia S, Sathananthan J, et al. Structural valve deterioration in transcatheter aortic bioprostheses: diagnosis, pathogenesis, and treatment. Struct Heart. 2023;7(3):100155. Published 2023 Apr 4. doi:10.1016/j.shj.2022.100155
- VARC-3 WRITING COMMITTEE, Généreux P, Piazza N, et al. Valve Academic Research Consortium 3: updated endpoint definitions for aortic valve clinical research. J Am Coll Cardiol. 2021;77(21):2717-2746. doi:10.1016/j.jacc.2021.02.038
- Trimaille A, Carmona A, Hmadeh S, et al. Transcatheter aortic valve durability: focus on structural valve deterioration. J Am Heart Assoc. 2025;14(13):e041505. doi:10.1161/JAHA.125.041505
- Thyregod HGH, Jørgensen TH, Ihlemann N, et al. Transcatheter or surgical aortic valve implantation: 10-year outcomes of the NOTION trial. Eur Heart J. 2024;45(13):1116-1124. doi:10.1093/eurheartj/ehae043
- Durand E, Eltchaninoff H, Tchetche D, et al. Predictors of outcomes of reintervention after transcatheter aortic valve replacement: FRANCE 2 and FRANCE TAVI registries. J Am Coll Cardiol. 2025;85(9):896-907. doi:10.1016/j.jacc.2024.11.048
- Jia Y, Maznyczka A, Boiago M, et al. Long-term durability of transcatheter aortic valves in patients with bicuspid aortic stenosis. Catheter Cardiovasc Interv. 2025;106(3):1746-1757. doi:10.1002/ccd.31742
- Tam DY, Wijeysundera HC, Naimark D, et al. Impact of transcatheter aortic valve durability on life expectancy in low-risk patients with severe aortic stenosis. Circulation. 2020;142(4):354-364. doi:10.1161/CIRCULATIONAHA.119.044559
- Guddeti RR, Bashir H, Seshiah P, El-Hangouche N, Kereiakes DJ, Garcia S. Redo-transcatheter aortic valve replacement: current evidence and procedural considerations. J Clin Med. 2025;14(18):6608. Published 2025 Sep 19. doi:10.3390/jcm14186608
- Lee GS, Tang G, Zaid S, Tam DY. The current state of redo transcatheter aortic valve replacement (TAVR) and limitations: why TAVR explant is important as the valve reintervention strategy. Ann Cardiothorac Surg. 2025;14(2):98-111. doi:10.21037/acs-2024-etavr-0149
- Blackman DJ, Aktaa S, Pickles D, et al. REdo transcatheter aortic VALVE implantation for the management of transcatheter aortic valve failure: design and rationale of the REVALVE study. Int J Cardiol. 2025;435:133400. doi:10.1016/j.ijcard.2025.133400
Clinical Topics: Cardiac Surgery, Cardiovascular Care Team, Invasive Cardiovascular Angiography and Intervention, Valvular Heart Disease, Aortic Surgery, Cardiac Surgery and VHD, Interventions and Structural Heart Disease
Keywords: Aortic Valve, Heart Valve Diseases, Transcatheter Aortic Valve Replacement