A Rare Type of Cardiac Metastatic Mass
A 54-year-old female patient with a history of cancer that had been treated 5 years prior presented to the hospital with chest pain and shortness of breath. Bedside echocardiography showed a pericardial effusion requiring a pericardial window for drainage. Positron emission tomography (PET) computed tomography (CT) scan showed mediastinal lymphadenopathy, and a transthoracic echocardiogram (TTE) showed the following (Figure 1).
Figure 1
Figure 1
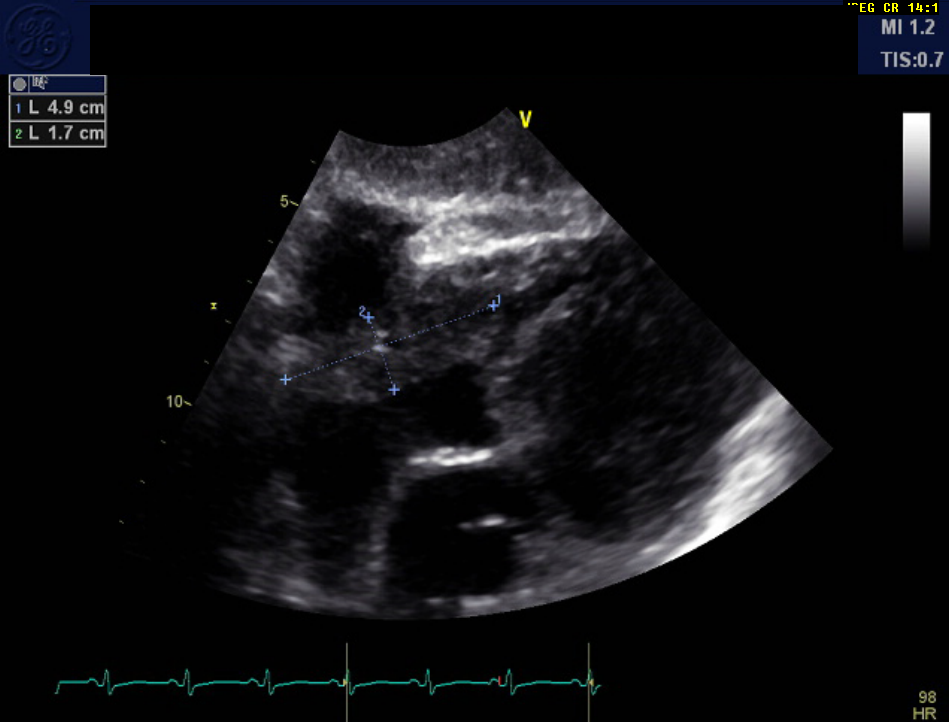
Which of the following is this mass least likely to represent?
Show Answer
The correct answer is: C. Metastatic cervical cancer
The patient was initially diagnosed with stage IIIB cervical cancer in 2012 at 49 years old. She was treated with chemotherapy and radiation (intracavitary brachytherapy using Ir-192 for a total dose of 84.3 Gy and high-dose-rate brachytherapy in 5 fractions of 6 Gy each. Her treatment was complicated by radiation enteritis and colitis leading to small bowel perforation for which she underwent multiple small bowel resections and a sigmoidectomy. She subsequently developed chronic, malabsorptive diarrhea due to short bowel syndrome and recurrent Clostridium difficile infections. Eventually, she underwent a diverting colostomy in January 2016.
The patient was followed without major complications for nearly 2 years until November 2014 when she presented with a painful left arm mass. PET CT revealed a soft tissue density with intense fludeoxyglucose (FDG) activity measuring up to 13 standardized uptake values, concerning for a neoplastic process. Pathology of a fine-needle biopsy specimen showed metastatic squamous cell carcinoma of cervical origin. She underwent local radiation therapy along with concurrent chemotherapy (cisplatin) January through March of 2015. A repeat, surveillance PET CT performed 5 months following treatment demonstrated interval shrinkage of the left upper arm mass (13 x 15 mm with non-focal FDG activity, standardized uptake values of 1.9), consistent with treated versus residual disease. Magnetic resonance imaging (MRI) of the left arm showed no residual disease. No metastatic disease was noted elsewhere.
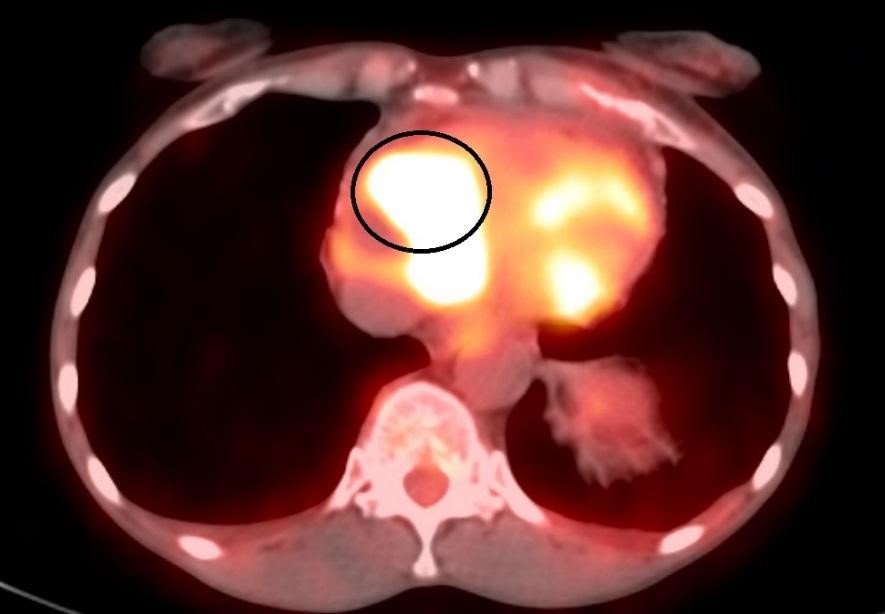
In June 2017, she returned to the hospital, this time with complaints of acute onset chest pain. According to medical records, she reported waking up from sleep with throbbing chest pain that was located under the left breast as well as nausea and vomiting. On TTE evaluation, the patient was found to have a pericardial effusion requiring pericardial window and drainage; 800 cc of bloody fluid was removed, and fluid analysis confirmed the presence of squamous cell carcinoma from cervical etiology. PET CT demonstrated new, prominent soft tissue within the right perivascular space adjacent to the ascending aorta with avid FDG activity, concerning for metastatic lymphadenopathy (Figure 2). Left 5th rib, distal humerus, and posterior vertebral body lesions were also detected. The patient was discharged from the hospital and was started on carboplatin/taxol/avastin with pegfilgrastim support 1 week later in the outpatient setting.
Figure 2
Figure 2
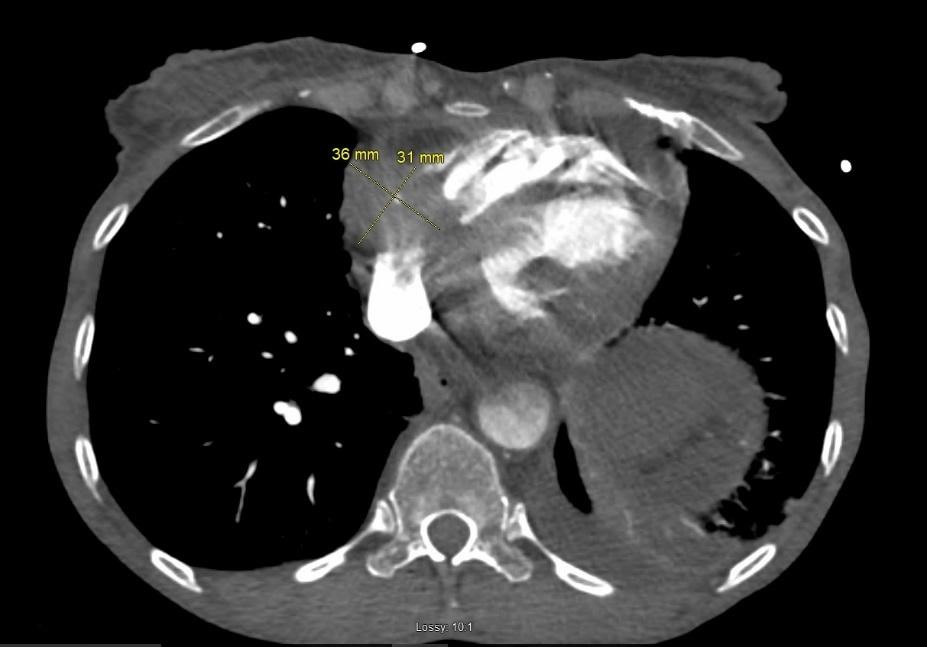
Three days after initiation of chemotherapy, the patient returned to the hospital with recurrent chest pain and shortness of breath. Troponin level was elevated (0.349). Cardiology was consulted and initiated colchicine therapy for pericarditis. Due to concern for a possible pulmonary embolism, a chest CT angiogram was performed, which was negative for a pulmonary embolism but revealed a prominent 36 x 31 mm soft tissue mass in the anterior mediastinum extending into the pericardium and the right atrioventricular groove (Figure 3).
Figure 3
Figure 3
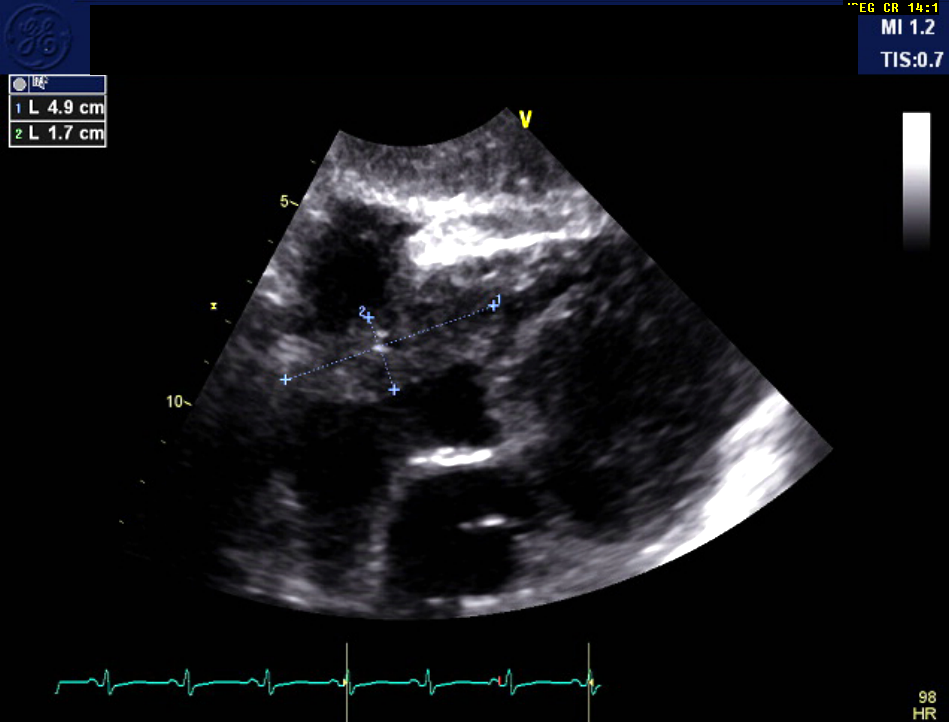
A formal echocardiogram was subsequently performed, which showed a large, 50 mm (maximal dimension), irregular-shaped mass extending from the pericardial space to the right ventricle (RV) and atria and resulting in moderate functional tricuspid stenosis. The mean gradient across the tricuspid valve was estimated at 5.4 mmHg. Due to the right atrial (RA) mass, assessment of RV function by tricuspid annular plane systolic excursion and S' was suboptimal, but RV function appeared grossly preserved (Figure 4).
Figure 4
Figure 4
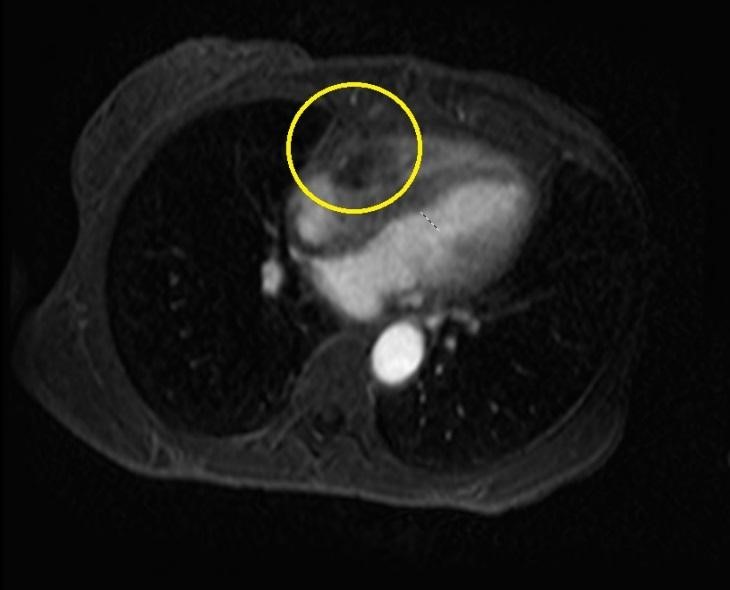
Cardiac MRI performed 4 days following admission revealed a small, anterior loculated pericardial effusion with a 41 x 40 mm heterogeneously enhancing soft tissue mass within the right atrioventricular groove, which appeared to be extracardiac with mass effect toward the RA and RV. Left ventricular ejection fraction was estimated at 53% (Figure 5).
Figure 5
Figure 5
The patient completed 6 cycles of chemotherapy (carboplatin/taxol/avastin) with pegfilgrastim support and had close oncologic follow-up. She was re-hospitalized, however, 4 months later with respiratory distress. The patient passed away on 12/8/17 from cardiac arrest secondary to acute hypoxic respiratory failure due to complications from underlying stage 4B cervical cancer.
Cardiac masses are rare and often incidental findings. They are broadly categorized as either neoplastic or non-neoplastic lesions. Although most non-neoplastic lesions are of intracardiac origin (i.e., thrombi), neoplastic lesions often represent metastases from extracardiac tumors. Metastases to the heart are approximately 40 times more common than primary cardiac tumors1 and occur anywhere from 1.5 to 21.8% among patients with malignancies, according to autopsy series. Cancers that most commonly metastasize to the heart include melanoma, lymphoma, leukemia, lung, and breast. Cases of gynecological malignancy metastasizing to the heart are extremely rare and seldom described in the literature. In reported cases, cardiac lesions of gynecologic origin were often detected only during an acute, clinical decompensation of the patient. In most instances, these lesions remain indolent and are rarely diagnosed before death.2
Extrapelvic metastases from recurrent cervical cancer typically occur in the lungs, liver, bones, and lymph nodes. The heart is rarely affected. In fact, the incidence of cardiac metastases of uterine cervix tumors in autopsy series is as low as 3-4%.1 More often, metastatic cardiac lesions are of lung or breast origin, presumably due to the relative proximity of these organs to the heart.
The three primary mechanisms of metastatic spread of neoplasms to the heart include retrograde lymphatic spread, hematogenous spread, and direct extension from adjacent viscera. In cases of cervical cancer, metastatic spread is often secondary to retrograde, regional lymphatic invasion through mediastinal lymphatic channels into the pericardium and epicardium. Metastatic implants on the pericardium can obstruct the lymphatic drainage from the visceral surface, which contains most of the lymphatic channels, resulting in a pericardial effusion. Implantation of cancer fragments in the myocardium is less frequent. When present, however, it is suggestive of hematogenous spread via the coronary arteries. Least common is the finding of metastasis within the cardiac chambers. Similar to myocardial metastases, intra-chamber implants occur via hematogenous spread, through the inferior vena cava and ultimately into the right cardiac chambers.1,3
The severity of clinical symptoms in patients with cardiac metastasis is variable, depending on the extent and localization within the heart. Cardiac metastasis may cause medical emergencies or, more often, remain unrecognized until identified incidentally on postmortem autopsy.1 In the 37 cases in which cardiac metastases were found before death, chest symptoms were the most prevalent 30 (81.5%) cases. Among those cases, the following symptoms were common:
- Sensation of dyspnea in 15 (50%)
- Dyspnea in 13 (43.3%)
- Chest pain in 10 (33.3%)
- Coughing in 6 (20.0%)
Early detection of cardiac metastasis is critical to improving prognosis. Diagnosis of cardiac tumors remains a diagnostic challenge, however, because they can often mimic non-neoplastic and occasionally native cardiac structures. Noninvasive imaging modalities have proven to be very effective in the evaluation and diagnosis of cardiac masses. Most cardiac masses are initially diagnosed by TTE. Echocardiography remains first line for cardiac-mass evaluation due to its widespread availability and lack of radiation exposure. Echocardiography not only provides detailed characteristics of the lesion, but also can be utilized to assess the presence of fluid in the pericardial space and any associated hemodynamic compromise. In certain cases, however, poor image quality with echocardiography limits its diagnostic value. CT and MRI are valuable tools for the visualization of inaccessible cardiac and thoracic structures and for the evaluation of extracardiac lesions. MRI can assess myocardial function comparably to echocardiography and renders excellent contrast resolution, allowing for the differentiation of tumor, myocardium, and pericardium. CT provides superior spatial resolution and renders the assessment of extracardiac structures.1
Pathophysiological changes that often occur in patients with cardiac metastasis include right-sided heart failure, increased coagulopathy, thrombocytopenia, and lung infarction from multiple pulmonary emboli, leading to respiratory failure. Although the prognosis of patients with metastatic heart disease is poor (nearing less than 6 months), tumor resection via open-heart surgery has been demonstrated in several cases with survival rates improving to 2 years.2 Current guidelines recommend aggressive local and systemic treatment to prevent hemodynamic instability and to achieve palliation of associated symptoms, irrespective of the primary malignancy.1
References
- Schawkat K, Hoksch B, Schwerzmann M, Puig S, Klink T. Diagnosis of cardiac metastasis from cervical cancer in a 33-year-old patient using multimodal imaging studies: a case report and literature review. Acta Radiol Short Rep 2014;3:2047981614530287.
- Okamoto K, Kusumoto T, Seki N, Nakamura K, Hiramatsu Y. A case of cardiac metastasis from uterine cervical carcinoma. Case Rep Obstet Gynecol 2015;2015:703424.
- Vilches E, Thekkumkattil A, De La Cruz Luque C, Sharim A, Villada G, Sancassani R. Right Ventricular Metastasis of Cervical Cancer Involving the Endomyocardium: A Case Report. Am J Respir Crit Care Med 2016;193:A6959.
