A 62-year-old male with a history of hypertension, hyperlipidemia, coronary artery disease with prior percutaneous coronary intervention to the left anterior descending artery, hypothyroidism, and stage III chronic kidney disease presented to cardiology clinic with several months of poor exercise tolerance, bilateral lower extremity edema, and fatigue.
He was initially diagnosed with acute pericarditis over 1 year ago, when he presented to an outside facility with fevers, chills, and pleuritic chest pain. Inflammatory markers were elevated, with C-reactive protein 157 mg/dL on admission. Extensive infectious workup including testing for Epstein-Barr virus, cytomegalovirus, ehrlichiosis, Legionella, Rickettsia, and Borrelia burgdorferi was negative. Antinuclear antibodies (ANA) was negative as well. Ultimately the etiology of pericarditis was labeled as idiopathic. He improved with a prolonged course of indomethacin 15 mg twice daily and colchicine 0.6 mg every other day, but 6 months he later returned with dyspnea and weight gain. Workup at the time was notable for cardiac magnetic resonance imaging (MRI) demonstrating a moderate pericardial effusion, circumferential pericardial thickening, diffuse delayed pericardial enhancement, no delayed myocardial enhancement, and diastolic septal bounce. Right heart catheterization showed equalization of diastolic filling pressures. He was diagnosed with constrictive pericarditis. He was continued on colchicine and started on a slow prednisone taper along with low dose furosemide. Despite an additional 3 months of medical therapy, his heart failure symptoms worsened, and he was referred to our facility for further management.
His medications at this visit included: colchicine 0.6 mg every other day, prednisone 40 mg daily, furosemide 40 mg daily, levothyroxine 150 mcg daily, amiodarone 200 mg daily, metoprolol succinate 50 mg daily, aspirin 81 mg daily, rivaroxaban 20 mg daily, and double strength sulfamethoxazole-trimethoprim 800 mg – 160 mg three times per week. His vital signs were normal, with heart rate 57 bpm, blood pressure 116/66 mmHg, oxygen saturation 97% on room air, and no pulsus paradoxus. Physical examination was notable for elevated jugular venous pressure, Kussmaul's sign, a pericardial knock, and 1+ pitting bilateral lower extremity edema. Laboratory evaluation showed elevated creatinine (1.40 mg/dL), elevated NTproBNP (289 pg/mL), normal troponin T (<0.010), normal ultra-sensitive C-reactive protein (0.9 mg/L), and normal Westergren sedimentation rate (5 mm/hr). Electrocardiogram demonstrated normal sinus rhythm with T wave inversions in the inferior leads.
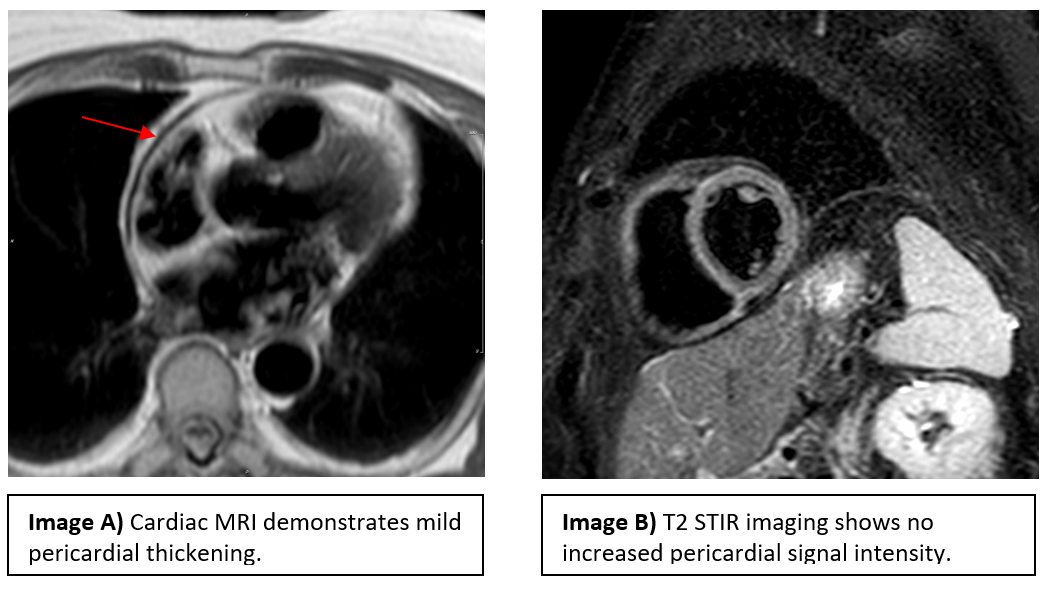
Cardiac MRI showed near concentric pericardial thickening, diastolic septal bounce, and mild respirophasic septal shift (Image A). There was no increased signal intensity of the pericardium on T2-weighted short-tau inversion recovery (STIR) sequences (Image B). Mild delayed enhancement of the pericardium adjacent to the basal anterior and lateral walls of the left ventricle was noted.
Left heart catheterization showed no angiographically significant coronary artery disease. Right heart catheterization demonstrated the following: mean RA pressure 17 mmHg, RV 40/20 mmHg, PA 36/24 mmHg with mean 26 mmHg, PCWP 24 mmHg, and LVEDP 22 mmHg. Cardiac index by Fick method was 4.17 L/min/m2.
The patient stated his symptoms were significantly impacting his quality of life and asked what else could be done.
The correct answer is: D. Refer for complete radical pericardiectomy
This patient's clinical presentation is most consistent with chronic idiopathic constrictive pericarditis, with symptoms refractory to medical management. Constrictive pericarditis is a rare complication of acute idiopathic pericarditis, developing in <1% of patients.1 The incidence of constriction is higher in patients with an identifiable etiology of pericarditis, with greatest risk seen following bacterial (especially tuberculous or purulent) pericarditis. The characteristic feature of constrictive pericarditis is a thickened, fibrotic, and/or calcified pericardium that restricts diastolic filling,2 leading to classic symptoms of right heart failure including fatigue, abdominal distension, and lower extremity edema.
In addition to this symptom constellation, the diagnosis of constriction is typically made using a combination of multi-modality imaging techniques. Transthoracic echocardiography is recommended in all patients with suspected constriction. Characteristic findings include septal bounce or ventricular septal shift with either medial e' > 8 cm/s or hepatic vein expiratory diastolic reversal ratio > 0.78.3 Abnormal mitral valve inflow velocity (E/A), a classic and reproducible feature of diastolic dysfunction, is expected as well.4 Cardiac computed tomography (CT) and cardiac MRI are indicated as second-level techniques to evaluate pericardial thickness, extent of pericardial involvement, presence of inflammation, and presence of calcifications. Heart catheterization is the gold standard to confirm diagnosis, demonstrating a rapid "y" descent on the atrial pressure wave form and the "square root sign" on ventricular pressure tracings (reflecting rapid early diastolic filling of the ventricles, followed by lack of further filling due to compression in mid and late diastole). Furthermore, loss of pericardial compliance leads to equalization of intracardiac diastolic pressures.5
Patients with newly diagnosed constrictive pericarditis and evidence of active inflammation on CT and/or MRI are typically managed with an initial trial of medical therapy including anti-inflammatory agents and diuretics as needed.6 If these conservative measures are unsuccessful, surgical pericardiectomy should be considered. Timing of surgery is critical; early operative intervention is technically challenging in the setting of active inflammation, whereas surgery after anti-inflammatory therapy may be more successful.7 The anterior pericardiectomy approach, characterized by phrenic to phrenic resection of the pericardium without the use of cardiopulmonary bypass, has been largely supplanted by the complete pericardiectomy.8 Complete resection is associated with superior functional outcome and survival when compared to anterior pericardiectomy.9
Returning to our patient, he started out with active inflammation with transient constriction and was treated medically for 1 year. His inflammation improved, as evidenced by normal inflammatory markers and only mild pericardial enhancement. However, the patient continued to feel unwell, with NYHA class II symptoms. Right heart catheterization confirmed persistent constrictive hemodynamics. In the setting of chronic symptomatic constriction despite resolution of inflammation, pericardiectomy was indicated. He underwent complete radical pericardiectomy, with pathology demonstrating organizing fibrous pericarditis with mild calcification. He was discharged from the hospital less than 2 weeks after surgery, with plan to continue a slow prednisone taper. At 1 month follow up, he was doing well with complete resolution of heart failure symptoms.
Constrictive pericarditis is a rare but potentially curable etiology of heart failure. Management requires a multifaceted approach involving anti-inflammatory medication, diuresis, and consideration of surgery in refractory cases. Serial inflammatory markers and multi-modality imaging can be useful in determining the potential role and optimal timing of surgery. Further study is needed to better delineate the management of anti-inflammatory medication after pericardiectomy, especially in the setting of pericardial enhancement at time of surgery.
References
- Adler Y, Charron P, Imazio M, et al. 2015 ESC Guidelines for the diagnosis and management of pericardial diseases: the task force for the diagnosis and management of pericardial diseases of the European Society of Cardiology (ESC) Endorsed by: The European Association for Cardio-Thoracic Surgery (EACTS). Eur Heart J 2015;36:2921-64.
- Chetrit M, Xu B, Kwon DH, et al. Imaging-guided therapies for pericardial diseases. JACC Cardiovasc Imaging 2020;13:1422-37.
- Welch TD, Ling LH, Espinosa RE, et al. Echocardiographic diagnosis of constrictive pericarditis: Mayo Clinic criteria. Circ Cardiovasc Imaging 2014;7:526-34.
- Chetrit M, Cremer PC, Klein AL. Imaging of diastolic dysfunction in community-based epidemiological studies and randomized controlled trials of HFpEF. JACC Cardiovasc Imaging 2020;13:310-26.
- Doshi S, Ramakrishnan S, Kumar Gupta S. Invasive hemodynamics of constrictive pericarditis. Indian Heart J 2015;67:175-82.
- Geske JB, Reddy Y. Pathophysiology and diagnosis of constrictive pericarditis. http://www.acc.org. Mar 13, 2017. Accessed 5/12/2020. https://www.acc.org/latest-in-cardiology/articles/2017/03/13/15/10/pathophysiology-and-diagnosis-of-constrictive-pericarditis.
- Cremer PC, Kumar A, Kontzias A, et al. Complicated pericarditis: understanding risk factors and pathophysiology to inform imaging and treatment. J Am Coll Cardiol 2016;68:2311-28.
- Unai S, Johnston DR. Radical pericardiectomy for pericardial diseases. Curr Cardiol Rep 2019;21:6.
- Chowdhury UK, Subramanian GK, Kumar AS, et al. Pericardiectomy for constrictive pericarditis: a clinical, echocardiographic, and hemodynamic evaluation of two surgical techniques. Ann Thorac Surg 2006;81:522-9.