A 21-year-old Black male, collegiate football player (defensive back), was referred for evaluation of an abnormal electrocardiogram (ECG) following an episode of atypical chest pain [Figure 1].
Figure 1: 12-lead ECG of a 21-year-old "genotype-positive, phenotype-negative" collegiate football player
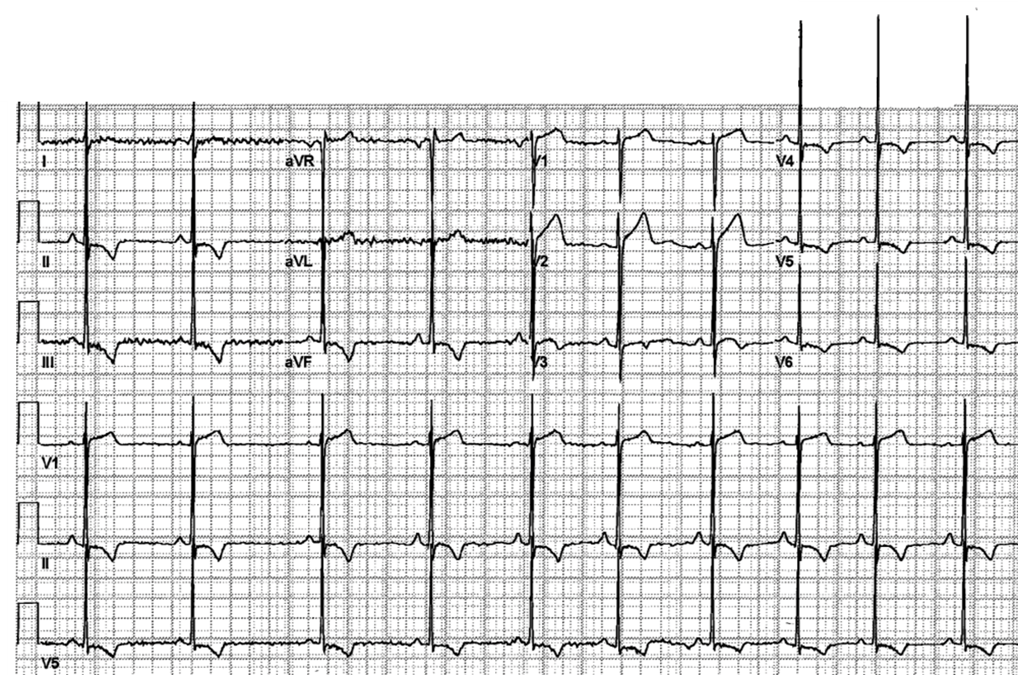 Figure 1: 12-lead ECG of a 21-year-old "genotype-positive, phenotype-negative" collegiate football player
Figure 1: 12-lead ECG of a 21-year-old "genotype-positive, phenotype-negative" collegiate football player
His chest pain symptoms, described as "sharp and brief", were noted at the time of an allergic flare. The patient reported no decline in exercise capacity or recent flu-like symptoms and had never experienced syncope or near syncope. His mother had been diagnosed with hypertrophic cardiomyopathy (HCM) in her 30's (MYH7 mutation) and required cardiac transplantation at age 51 for refractory heart failure symptoms. Her son was found to be a carrier of the same MYH7 gene variant (c.2543 A> p.Glu848Gly [E848G]). There was no personal or family history of sickle cell disease/trait.
Coronary computed tomography (CT) angiography demonstrated no anomalous coronary artery origins, myocardial bridge, or obstructive atherosclerotic lesions. Transthoracic echocardiography (TTE) revealed normal left ventricular (LV) chamber dimensions and wall thickness (septum and posterior wall 0.7 cm) with a left ventricular ejection fraction (LVEF) of 69%. LV diastolic function indices were normal. There was no evidence of mitral regurgitation, systolic anterior motion (SAM), or left ventricular outflow tract (LVOT) gradient at rest and with Valsalva. Cardiac magnetic resonance imaging (MRI) confirmed normal LV wall thickness measures with no delayed gadolinium enhancement evident. During treadmill exercise testing, the patient completed 14.5 minutes on a Bruce protocol achieving a heart rate of 166/minute without symptoms, diagnostic ST segment changes, or ventricular dysrhythmias. The blood pressure (BP) response to exercise was normal. A repeat TTE performed the following year showed no changes, and 48-hour Holter monitor identified no ventricular tachycardia.
The correct answer is: D. Allowance for continued training and competitive sports participation is reasonable once provisions for ongoing cardiac re-evaluation are in place.
Identification of cardiovascular disorders in athletes for which intense training and competition are best avoided can be a challenging and at times perilous endeavor. A divergence of opinion previously existed between European and United States (ACC/AHA) recommendations for competitive sporting participation in the genotype-positive, phenotype-negative athlete.1,2 European experts initially advocated sporting restrictions for such individuals while provision for full participation was allowed in the US guidelines. The disparity related to the publication timelines of respective expert documents with a number of observational longitudinal studies showing the benign nature of this cohort following publication of the European guidelines. Evolving data, showing a lack of incurred risk in such individuals, culminated in the 2019 European recommendations which also permit full sporting participation.3 A more challenging nuance is the clear and precise definition of the term "phenotypic expression." Phenotype refers to the expression of the disease (derived from the Greek words "phainein" meaning "appear" and "typos" which means "type"), which could reasonably be inferred to imply depolarization and depolarization changes on the ECG. However, limited data also suggest that ECG changes without accompanying morphological changes on cardiac imaging has a benign course.4 Caselli and Pelliccia5 have recently examined the issue of the genotype-positive, phenotype-negative, ECG-positive athlete and proposed an algorithmic approach to determine sports eligibility [Figure 2].
Figure 2: Proposed management algorithm for athletes with a genetic mutation for hypertrophic cardiomyopathy (from Caselli and Pelliccia5 with permission from the publisher).
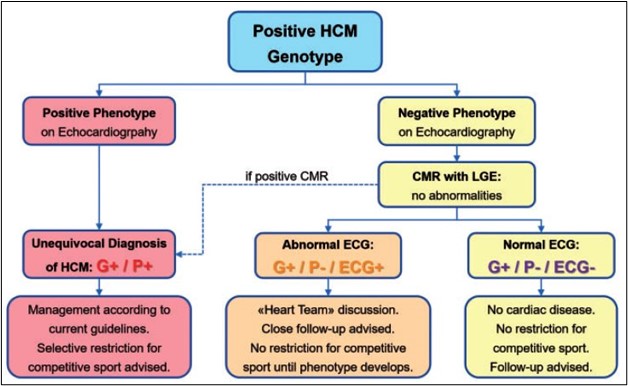 Figure 2: Proposed management algorithm for athletes with a genetic mutation for hypertrophic cardiomyopathy (from Caselli and Pelliccia5 with permission from the publisher).
Figure 2: Proposed management algorithm for athletes with a genetic mutation for hypertrophic cardiomyopathy (from Caselli and Pelliccia5 with permission from the publisher).
In the present case, we discussed the potential risks and uncertainties of continued training and competitive participation, importance of symptom recognition/reporting, and requirement for continued disease surveillance with the athlete, his family, coaches, and training staff. However, we did not mandate sporting restriction. The student-athlete was given a one-year clearance to play with the requirement for annual follow-up with cardiac imaging. He has played without incident or progression of LVH over the past 3 years.
Answer A is incorrect. The patient's ECG was not consistent with the "benign repolarization pattern of Black athletes". The ECG pattern frequently recognized in those of African or Afro-Caribbean descent is characterized by T wave inversion (TWI) in leads V1 through V4 preceded by J-point elevation and convex ("domed") ST-segment elevation [Figure 3].6,7
Figure 3: ECG showing the typical features of the "benign repolarization variant of the Afro-Caribbean Athlete"2,3
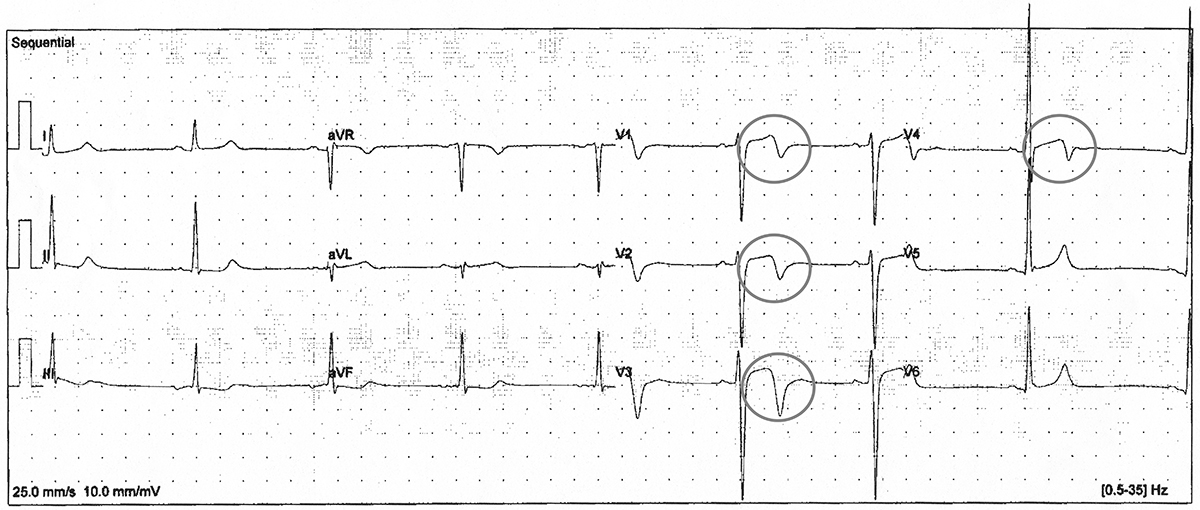 Figure 3: ECG showing the typical features of the "benign repolarization variant of the Afro-Caribbean Athlete"2,3
Figure 3: ECG showing the typical features of the "benign repolarization variant of the Afro-Caribbean Athlete"2,3
While athletes occasionally exhibit diffuse TWI in the absence of underlying structural heart disease, lateral TWI should always be considered pathologic until proven otherwise.6 Such findings may represent the earliest expression of hypertrophic cardiomyopathy (HCM) in apparently healthy athletes and mandate regular clinical follow-up with serial cardiac imaging, even when initial testing is normal.
Answer B is incorrect. There is limited evidence that continued participation in rigorous physical training will accelerate expression of an HCM phenotype.
Many individuals with pathologic TWI will not be shown to harbor occult pathology despite comprehensive evaluation. Pre-emptive disqualification may unnecessarily deprive such individuals of the physical, emotional, and potential financial benefits of participation in sports activities. While intense physical training has been shown to accelerate disease expression in certain conditions such as arrhythmogenic cardiomyopathy (AC) and myocarditis, accelerated remodeling has not been demonstrated in individuals with HCM.
Answer C is incorrect. Disqualification from competitive sports is not mandated for all athletes who are HCM genotype-positive, phenotype-negative.
With more inclusive athletic screening programs, cardiac imaging, and genetic testing services, the sports cardiologist will inevitably encounter individuals with HCM who are "genotype-positive, phenotype-negative". The decision to permit ongoing physical training and competition must be considered on a case by case basis. The most recent ACC/AHA guidelines addressed this distinctive population: "At present, the risk for sudden death in gene-positive, phenotype-negative family members appears to be extremely low and likely no different from the risk in the general population of the same age without heart disease"..."Participation in competitive athletics for asymptomatic, genotype-positive HCM patients without evidence of LV hypertrophy by 2-dimensional echocardiography and CMR is reasonable, particularly in the absence of a family history of HCM-related sudden death [Class IIa; Level of Evidence C]."2 Caselli and Pelliccia5 have proposed a management algorithm for athletes identified with a pathologic or likely pathologic HCM gene variant identified during family screening [Figure 2].
Answer E is incorrect. Genetic testing is not recommended as part of the evaluation of young, asymptomatic athlete with TWI.
The diagnostic yield from genetic testing in asymptomatic young athletes with TWI and normal echocardiographic studies has not been shown to add significantly to the clinical evaluation, making it of negligible value in routine clinical practice.8
References
- Pelliccia A, Fagard R, Bjornstad HH, et al. Recommendations for competitive sports participation in athletes with cardiovascular disease: a consensus document from the study group of sports cardiology of the working group of cardiac rehabilitation and exercise physiology and the working group of myocardial and pericardial diseases of the European Society of Cardiology. Eur Heart J 2005;26:1422-45.
- Maron BJ, Udelson JE, Bonow RO, et al. Eligibility and disqualification recommendations for competitive athletes with cardiovascular abnormalities: Task Force 3: hypertrophic cardiomyopathy, arrhythmogenic right ventricular cardiomyopathy and other cardiomyopathies, and myocarditis: a scientific statement from the American Heart Association and American College of Cardiology. J Am Coll Cardiol 2015; 66:2362-71.
- Pelliacia A, Solberg EE, Papadakis M, et al. Recommendations for participation in competitive and leisure time sport in athletes with cardiomyopathies, myocarditis, and pericarditis: position statement of the Sport Cardiology Section of the European Association of Preventive Cardiology (EAPC). Eur Heart J 2019;40:19-33.
- Pelliccia A, Lemme E, Maestrini V, et al. Does sports participation worsen the clinical course of hypertrophic cardiomyopathy? Circulation 2018;137:531-33.
- Caselli S, Pelliccia, A. The electrocardiogram and the phenotypic expression of hypertrophic cardiomyopathy. Eur Heart J 2019;40:982-85.
- Papadakis M, Carre F, Kervio G, et al. The prevalence, distribution, and clinical outcomes of electrocardiographic repolarization patterns in male athletes of African/Afro-Caribbean origin. Eur Heart J 2011;32:2304-13.
- Sharma S, Drezner JA, Baggish A, et al. International recommendations for electrocardiographic interpretation in athletes. J Am Coll Cardiol 2017;69:1057-75.
- Sheikh N, Papadakis M, Wilson M, et al. Diagnostic yield of genetic testing in young athletes with T-wave inversion. Circulation 2018;138:1184-94.



