The One-Year Results of the JET-ISR Study
Quick Takes
- Jetstream™ atherectomy is safe and effective in debulking femoropopliteal in-stent restenotic tissue.
- Jetstream™ atherectomy yields statistically lower target lesion revascularization than balloon angioplasty in the treatment of femoropopliteal in-stent restenosis.
- In general, debulking of in-stent restenosis is effective, but drug coated balloons, VIABAHN® covered stents, and combination therapy of debulking and drug coated balloons appear numerically superior to debulking alone. No head-to-head comparison is available for Jetstream™ atherectomy in in-stent restenosis treatment.
Introduction
In-stent restenosis (ISR) of the femoropopliteal artery (FP) following self-expanding bare metal stent ranges from 19 to 37% at 1 year.1-4 Despite new bare metal stent designs (Supera™ [Abbott], Biomimics 3D® Vascular Stent System [Veryan], MISAGO® stent [Terumo]), or drug eluting stents ( Zilver® PTX® [Cook Medical], Eluvia™ [Boston Scientific]) the loss of patency continues to be seen in about 15 to 20% of FP stented patients on 1-year follow up.5-8
Re-intervention on restenotic FP lesions using balloon angioplasty (PTA) alone has a high rate of recurrence of restenosis and repeat target lesion revascularization (TLR).9 Restenotic tissue debulking in FP ISR appears to reduce or delay the need for repeat revascularization and bail-out stenting. In this expert analysis we discuss Jetstream™ atherectomy (JA) as a debulking technique in treating FP ISR.
Jetstream™ Atherectomy in FP-ISR: Pre-JET-ISR
The application of JA was initially reported by Beschorner et al.10 on 33 patients (40 infrainguinal ISR lesions) treated with the Pathway PV Atherectomy System, the predecessor of the current Jetstream™ system. Primary patency was disappointingly 33 % after 1 year and 25 % at 2 years. Distal embolization occurred in 6% of lesions and bail out stenting was needed in one lesion (3%). Since then, the Pathway System was improved upon with high cutting capability and stronger aspiration system.
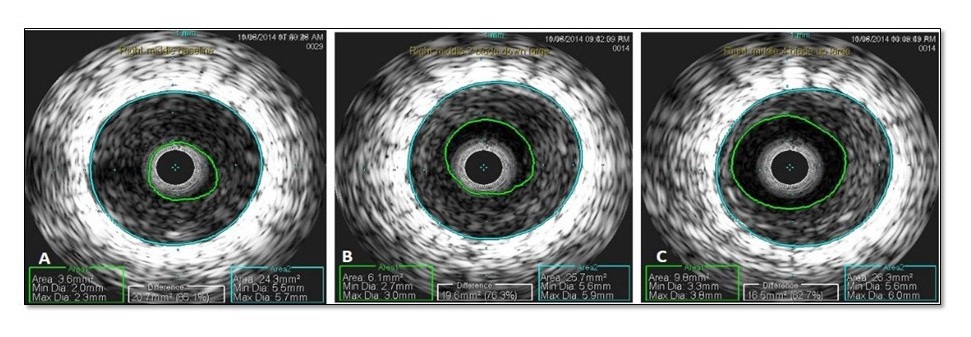
Preclinical data was then conducted using JA on FP ISR in overstretched porcine arteries. Data indicated that 2 blade-down runs (each run is from proximal to distal part of the lesion) followed by 2 blade-up runs lead to optimal tissue excision with maximum minimal luminal area gain by the Jetstream™ device11 (Figure 1). This was later validated in a small number of patients with FP ISR.12 Also, preclinical data did not indicate stent disruption which was examined by electron microscopy and high dose radiographs.11
Figure 1
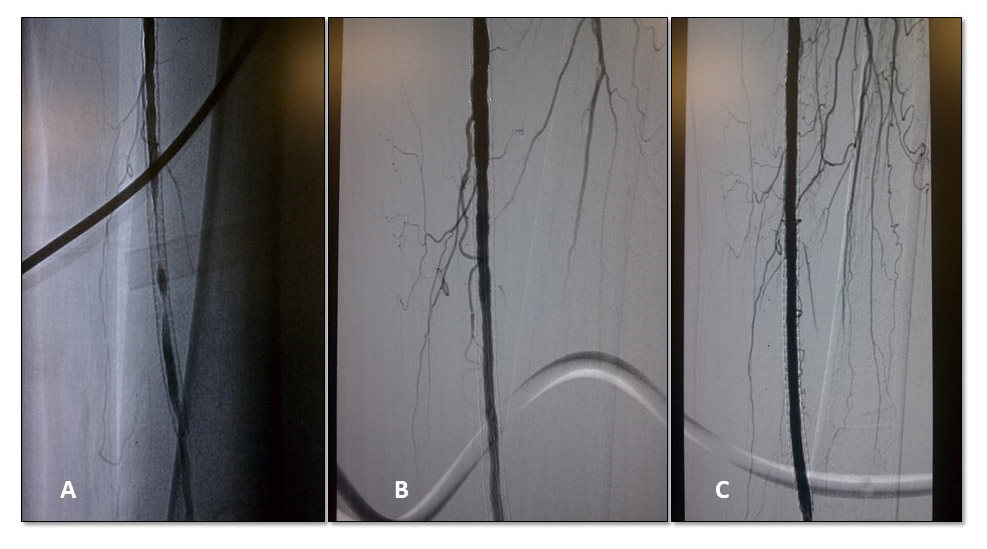
The Jetstream™ -ISR registry was a prospective, pilot registry designed to validate the safety of patients treated with JA for FP ISR.13 In this feasibility study with angiographic core laboratory evaluating stent integrity post-JA, 29 patients (32 limbs) were included. All patients were treated with adjunctive balloon angioplasty (Figure 2). Total lesion length was 17.4 ± 13.1 cm and total treated length 19.5 ± 12.9 cm. Distal embolization occurred in 9.4%. Embolic filter protection was used in half the limbs and macrodebris was noted in 12.5% of filters. There were no new stent fractures or deformities (n=24) as reported by Core Lab. TLR occurred in 13.8% and 41.4% of patients at 6-month and 1-year respectively. Patency rate was 72% at 6 months.
Figure 2
Jetstream™ Atherectomy in FP ISR: The JET-ISR trial
The JET-ISR study14 was a prospective study conducted in 10 centers in the United States and included 60 FP-ISR patients. Lesion length was 19.9±13.5 cm; 33 (55%) were chronic total occlusion (CTO). No drug eluting balloons or stents were allowed. Similar to the Jetstream™ ISR study, core laboratory evaluated stent integrity at baseline, after JA and after final PTA. The primary endpoint was TLR at 6 months with bailout stenting considered as TLR. Secondary endpoints included TLR (without bailout stenting) and clinical patency (no restenosis or TLR) at 1 year.
No stent fracture or deformities were noted confirming early data from the Jetstream™- ISR. Bailout stenting occurred in 10% of limbs. At 6-month and 1-year, freedom from TLR were 79.3% (95% CI 68.9% to 89.8%) and 60.7% (95% CI 47.8% to 73.6%), respectively, with bailout stenting considered as TLR; and 89.3% (95% CI 81.2% to 97.4%) and 66.8% (95% CI 54.3% to 74.2%) respectively when bailout stenting not considered as TLR. Clinical patency rates at 6 months and 1 year were 77.5% (31/40) and 51.7% (15/29), respectively. When proportional TLR rate of 21.4% (95% CI upper bound=32.4%) was compared to a prespecified historic control derived from a metanalysis of the PTA arms of EXCITE-ISR,15 RELINE,16 and FAIR17 (performance goal was set at 37.9% at 6-month follow-up with bailout stenting considered as TLR), JA showed a statistically lower TLR rate at 6-month than PTA (p=0.007).
A meta-analysis of the Jetstream™ ISR and JET-ISR was recently published.18 A total of 92 lesions were treated in 89 patients. Proportional TLR was 21.2%. When this was compared to a performance goal of 37.9% at 6 months with PTA alone, the TLR rate with JA was significantly lower than the TLR rate set for the historic control. Distal embolization occurred in 12.0% (filters used in 76.1% of all cases) and bailout stenting was 8.7%. This data was consistent with laser debulking using EXCImer as seen in the EXCITE-ISR trial.
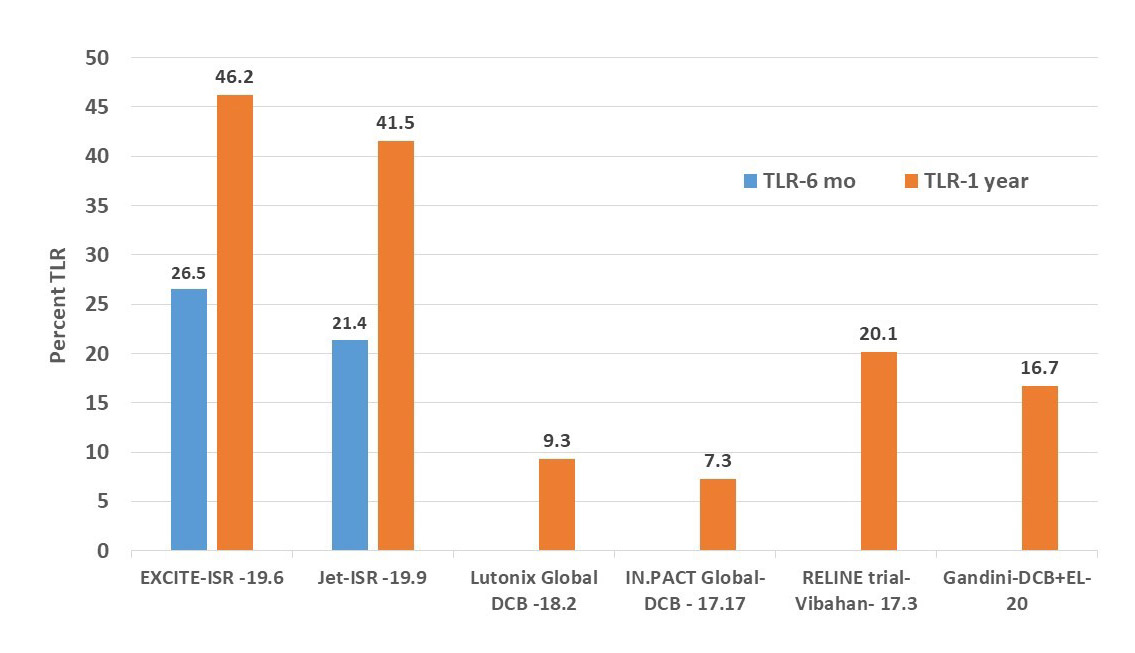
Despite a statistically lower TLR rate than PTA, debulking with JA appears to have a continued higher TLR rate than DCB or VIABAHN® stents (Figure 3).
Figure 3
Jetstream™ Atherectomy with Drug Coated Balloon in Treating FP ISR
In 48 critical limb ischemia patients with chronic SFA in-stent occlusion19 randomly assigned to laser atherectomy with DCB (n=24) versus DCB alone (n=24), patency at 12 months was 66.7% with laser and adjunctive DCB versus 37.5% in the DCB arm. The JET-SCE study20 was a retrospective analysis that included all comers including de novo disease, restenosis, and in-stent restenosis. In this study, 50 patients received JA alone with adjunctive PTA and 25 patients JA with adjunctive DCB. Freedom from TLR was significantly higher with JA and DCB versus JA and PTA at 12 months (94.7% vs. 68.0%, p = 0.002) and 16 months (94.4% vs. 54%; p = 0.002). The number of FP ISR in this study was small and therefore at this time more data is needed.
Conclusion
JA appears to be safe in treating FP ISR. Stent integrity seems to be preserved but advanced stent fractures (III and IV) at baseline have been excluded from the JET-ISR. Distal embolization remains a problem despite the built-in aspiration system in the Jetstream™ device. Combination therapy is promising with performing debulking and DCB for FP ISR lesions when compared to debulking alone or DCB alone. However, multicenter, randomized data are needed to have level A evidence before the combination therapy can be routinely recommended.
References
- Schillinger M, Sabeti S, Loewe C, et al. Balloon angioplasty versus implantation of nitinol stents in the superficial femoral artery. N Engl J Med 2006;354:1879-88.
- Laird JR, Katzen BT, Scheinert D, et al. Nitinol stent implantation versus balloon angioplasty for lesions in the superficial femoral artery and proximal popliteal artery: twelve-month results from the RESILIENT randomized trial. Circ Cardiovasc Interv 2010;3:267-76.
- Schillinger M, Sabeti S, Dick P, et al. Sustained benefit at 2 years of primary femoropopliteal stenting compared with balloon angioplasty with optional stenting. Circulation 2007;115:2745-9.
- Laird JR, Katzen BT, Scheinert D, et al. Nitinol stent implantation vs. balloon angioplasty for lesions in the superficial femoral and proximal popliteal arteries of patients with claudication: three-year follow-up from the RESILIENT randomized trial. J Endovasc Ther 2012;19:1-9.
- Bhatt H, Kovach R, Janzer S, George JC. SUPERA stent outcomes in Above-The-Knee IntervEntions: effects of COMPression and ELongation (SAKE-COMPEL) Sub-study. Cardiovasc Revasc Med 2018;19:512-15.
- Zeller T, Gaines PA, Ansel GM, Caro CG. Helical centerline stent improves patency: two-year results from the randomized mimics trial. Circ Cardiovasc Interv 2016;9:e002930.
- Gouëffic Y, Sauguet A, Desgranges P, et al. A polymer-free paclitaxel-eluting stent versus a bare-metal stent for de novo femoropopliteal lesions: The BATTLE Trial. JACC Cardiovasc Interv 2020,13:447-57.
- Gray WA, Keirse K, Soga Y, et al. A polymer-coated, paclitaxel-eluting stent (Eluvia) versus a polymer-free, paclitaxel-coated stent (Zilver PTX) for endovascular femoropopliteal intervention (IMPERIAL): a randomised, non-inferiority trial. Lancet 2018;392:1541-51.
- Shammas NW, Jones-Miller S, Lemke J. Meta-analysis-derived benchmarks of patency and target lesion revascularization of percutaneous balloon angioplasty from prospective clinical trials of symptomatic femoropopliteal in-Stent restenosis. J Vasc Interv Radiol 2016;27:1195-203.
- Beschorner U, Krankenberg H, Scheinert D, et al. Rotational and aspiration atherectomy for infrainguinal in-stent restenosis. Vasa 2013;42:127-33.
- Shammas NW, Aasen N, Bailey L, Budrewicz J, Farago T, Jarvis G. Two blades-up runs using the Jetstream navitus atherectomy device achieve optimal tissue debulking of nonocclusive in-stent restenosis: observations from a porcine stent/balloon injury model. J Endovasc Ther 2015;22:518-24.
- Shammas NW, Shammas GA, Aasen N, Jarvis G. Number of blades-up runs using Jetstream XC atherectomy for optimal tissue debulking in patients with femoropopliteal artery in-stent restenosis. J Vasc Interv Radiol 2015;26:1847-51.
- Shammas NW, Shammas GA, Banerjee S, Popma J, Mohammad A, Jerin M. Jetstream rotational and aspiration atherectomy in treating in-stent restenosis of femoropopliteal arteries: results of the JETSTREAM™ -ISR Feasibility Study. J Endovasc Ther 2016;23:339-46.
- Shammas NW, Petruzzi N, Henao S, et al. Jetstream atherectomy for the treatment of in-stent restenosis of the femoropopliteal segment: one-year results of the JET-ISR Study. J Endovasc Ther 2021;28:107-16.
- Dippel EJ, Makam P, Kovach R, et al. Randomized controlled study of excimer laser atherectomy for treatment of femoropopliteal in-stent restenosis: initial results from the EXCITE ISR trial (EXCImer Laser Randomized Controlled Study for Treatment of FemoropopliTEal In-Stent Restenosis). JACC Cardiovasc Interv 2015;8:92-101.
- Bosiers M, Deloose K, Callaert J, et al. Superiority of stent-grafts for in-stent restenosis in the superficial femoral artery: twelve-month results from a multicenter randomized trial. J Endovasc Ther 2015;22:1-10.
- Krankenberg H, Tübler T, Ingwersen M, et al. Drug-coated balloon versus standard balloon for superficial femoral artery in-stent restenosis: the randomized Femoral Artery In-Stent Restenosis (FAIR) Trial. Circulation 2015;132:2230-6.
- Shammas NW, Jones-Miller S, Shammas GA, Shammas WJ. Jetstream atherectomy in treating femoropopliteal in-stent restenosis: meta-analysis of the JETSTREAM -ISR and JET-ISR Trials. J Invasive Cardiol 2020;32:289-94.
- Gandini R, Del Giudice C, Merolla S, Morosetti D, Pampana E, Simonetti G. Treatment of chronic SFA in-stent occlusion with combined laser atherectomy and drug-eluting balloon angioplasty in patients with critical limb ischemia: a single-center, prospective, randomized study. J Endovasc Ther 2013;20:805-14.
- Shammas NW, Shammas GA, Miller SJ, et al. Long-term outcomes with Jetstream atherectomy with or without drug coated balloons in treating femoropopliteal arteries: a single center experience (JET-SCE). Cardiovasc Revasc Med 2018;19:771-77.
Clinical Topics: Cardiac Surgery, Invasive Cardiovascular Angiography and Intervention, Noninvasive Imaging, Stable Ischemic Heart Disease, Aortic Surgery, Cardiac Surgery and SIHD, Statins, Interventions and Imaging, Echocardiography/Ultrasound, Chronic Angina, Vascular Medicine
Keywords: Prospective Studies, Drug-Eluting Stents, Toll-Like Receptor 9, Retrospective Studies, Feasibility Studies, Coronary Restenosis, Follow-Up Studies, Atherectomy, Angioplasty, Balloon, Biometry, Femoral Artery, Registries, Microscopy, Electron, Ultrasonography, Interventional, Ischemia, Phenobarbital, Pharmaceutical Preparations
< Back to Listings