For Best Results in ACS PCI: Treat Upstream With P2Y12 Inhibitors
Quick Takes
- Pre-treatment with a P2Y12 inhibitor reduces cardiovascular events in the ACS population.
- The benefit of pre-treatment is time dependent and increases with the length of time between pre-treatment and the cath lab.
- In healthcare systems where NSTEMI patients are not routinely taken to the cath lab within the first few hours of presentation early treatment is an important component of NSTEMI management.
- In STEMI patients an early pre-treatment strategy reduces stent thrombosis, and considering >95% of STEMI patients undergo PCI, all patients with STEMI should receive early treatment with a P2Y12 inhibitor.
The Benefit of P2Y12 Inhibitor Pre-treatment
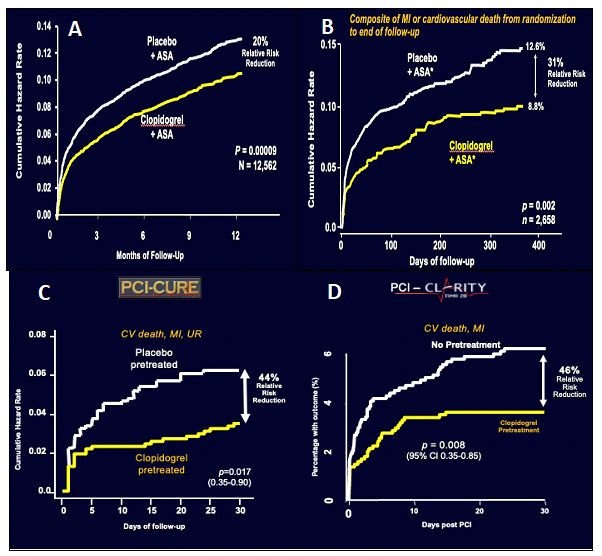
The benefit of P2Y12 inhibition in addition to acetylsalicylic acid (ASA) in the non-ST segment elevation acute coronary syndromes (NSTE-ACS) population was initially shown in the CURE trial where the addition of clopidogrel to ASA resulted in a significant 20% reduction in cardiovascular (CV) death, myocardial infarction (MI), or stroke at 12 months (RR 0.80 CI 0.72-090; P<0.001, NNT=48) (Figure 1A).1 In the percutaneous coronary intervention (PCI) era, one of the criticisms of the CURE data is that a large portion of the patients in the trial were medically managed. However, when you look at the population of 2,658 CURE patients who underwent PCI, there was a further reduction in CV events with a relative risk reduction of 31% in the composite outcome of CV death or MI (P= 0.002) and a relative risk reduction of 44% when adding urgent target vessel revascularization to that composite outcome at 30 days (CI 0.35-0.9; P=0.017) (Figure 1B,C).2
In this trial, a key feature was that P2Y12 inhibition was started early after randomization, before coronary anatomy had been defined, which has resulted in the clinical practice of pre-treatment with both ASA and clopidogrel.
Figure 1
The utility of pre-treatment in the ST-elevated myocardial infarction (STEMI) population was investigated in the PCI-CLARITY trial. This trial showed that when pre-treated with P2Y12 inhibition, STEMI patients had a 46% relative risk reduction in CV death or MI at 30 days (CI 0.35-0.85; P= 0.08) (Figure 1D).3 This is in keeping with the relative risk reductions favoring pre-treatment with clopidogrel in the non-ST-elevation myocardial infarction (NSTEMI) population outlined in CURE.1,3 Further data supporting pre-treatment can be seen in the meta-analysis by Bellemain-Appaix et al. published in JAMA in 2012 which included data from CURE and PCI-CLARITY as well as other select randomized controlled clinical trials (RCTs) and observational studies. This analysis showed a significant reduction in major coronary events when patients were pre-treated with a P2Y12 inhibitor.4 This held true for both meta-analysis of RCT data (OR 0.77; 0.66-0.89; P < 0.001)4 as well as meta-analysis of observational data (OR 0.70; CI 0.56-0.88; P=0.002)4 outlining a consistent benefit with pre-treatment. When looking at this data, it appears clear and unequivocal that pre-treatment is beneficial in patients with ACS.
The Importance of Timing of Pre-Treatment
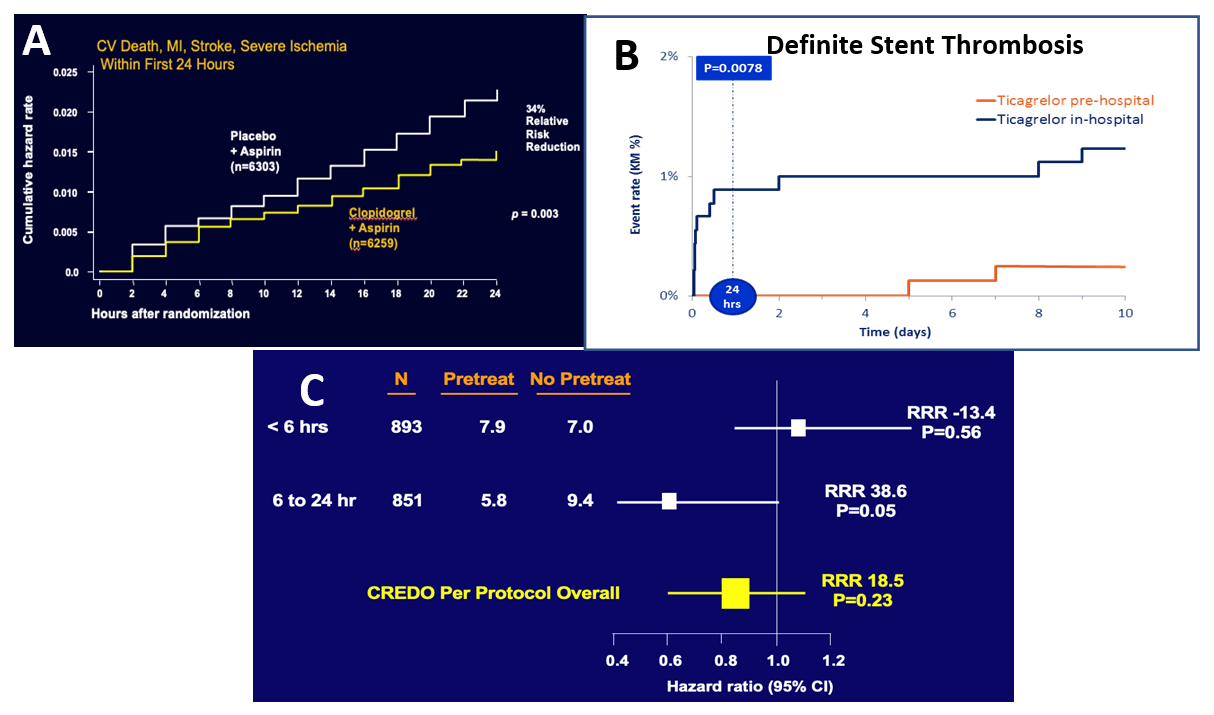
Timing of pre-treatment has important implications for CV outcomes. Upon a closer look at the CURE data, an important finding is that the treatment effect of clopidogrel emerged early after the loading dose had been administered. As you can see in the data displayed in Figure 2A, there is already clear separation in the Kaplan-Meyer curves by 6-8 hours and by 24 hours there is a clear significant benefit with a relative risk reduction of 34% (P= 0.003) with clopidogrel pre-treatment for preventing the composite outcome of CV death, MI, stroke, or severe ischemia.5 This is an important consideration when deciding on whether to give a loading dose of P2Y12 inhibitor to a patient who may not be going to the cath lab immediately.
Implications regarding timing of pre-treatment with a P2Y12 inhibitor in the STEMI population can be seen in the ATLANTIC trial comparing pre-hospital administration of ticagrelor with in-hospital administration. While there was no difference between the groups for the non-clinical primary outcomes of ST-elevation resolution and TIMI grade III flow in the culprit vessel, there was a significant decrease in the rates of definite stent thrombosis in the pre-hospital treatment arm (Figure 2B).6 This is a clinical outcome with significant impact on patients that should be used to guide our treatment strategy in the STEMI patient population.
Figure 2
Finally, one of the most important and relative points elucidated in 2021 is that the benefit of pre-treatment is likely to be time dependent, meaning that an oral loading dose strategy may take some time to work. This is shown in the CREDO trial data where benefits of pre-treatment with P2Y12 inhibitor in reducing the composite outcome of death, MI or urgent target vessel revascularization were seen in the population of patients who received a loading dose between 6-24 hours pre-PCI, however in the population of patients who received their loading dose <6 hours prior to PCI there was no benefit (Figure 2C).7 When you think about this from a biological standpoint this makes sense as it takes time for drug absorption and metabolization. A follow up point in this regard applies to the ACCOAST trial which has been used to amend guidelines in favor of not pre-treating with P2Y12 inhibitors. Although the trial did not show a significant difference in its primary efficacy outcome with pre-treatment (CV death, MI, stroke, UR, GP IIb/IIIa bailout),8 the median time from pre-treatment to PCI was only 4 hours. We already knew from CREDO data that patients who go to the cath lab this early are unlikely to benefit from pre-treatment.7,8 However, the ACCOAST trial does not address the value of pre-treatment for longer time intervals. We also know that patients with a longer time from presentation to PCI have a greater benefit from pre-treatment, which is a critical point in healthcare settings without 24/7 cath lab access for NSTEMI patients. Considering this, we would urge cardiologists to think about the patient in front of them, their individual risk, and consider the time that it would take that patient to get to the cath lab when deciding on pre-treatment.
Conclusions
The totality of data from RCTs and observational studies demonstrate that pre-treatment reduces CV events. This benefit of pre-treatment appears to be time dependent in that the longer the period between pre-treatment and the cath lab, the greater the benefit. Conversely, in patients going to the cath lab very early (i.e., within 4 hours) there may be no benefit to pre-treatment. Therefore, in healthcare systems where NSTEMI patients are not routinely taken to the cath lab within the first few hours of presentation, early treatment with P2Y12 inhibitor is an important component of NSTEMI management. In STEMI patients, an early pre-treatment strategy significantly reduces stent thrombosis and because >95% of STEMI patients undergo PCI, all patients with STEMI should receive early treatment with a P2Y12 inhibitor.
References
- Yusuf S, Zhao F, Mehta SR, Chrolavicius S, Tognoni G, Fox KK. Effects of clopidogrel in addition to aspirin in patients with acute coronary syndromes without ST-segment elevation. N Engl J Med 2001;345:494-502.
- Mehta SR, Yusuf S, Peters RJ, et al. Effects of pretreatment with clopidogrel and aspirin followed by long-term therapy in patients undergoing percutaneous coronary intervention: the PCI-CURE study. Lancet 2001;358:527-33.
- Sabatine MS, Cannon CP, Gibson CM, et al. Effect of clopidogrel pretreatment before percutaneous coronary intervention in patients with ST-elevation myocardial infarction treated with fibrinolytics: the PCI-CLARITY study. JAMA 2005;294:1224-32.
- Bellemain-Appaix A, O'Connor SA, Silvain J, et al. Association of clopidogrel pretreatment with mortality, cardiovascular events, and major bleeding among patients undergoing percutaneous coronary intervention: a systematic review and meta-analysis. JAMA 2012;308:2507-16.
- Yusuf S, Mehta SR, Zhao F, et al. Early and late effects of clopidogrel in patients with acute coronary syndromes. Circulation 2003;107:966-72.
- Montalescot G, Lassen JF, Hamm CW, et al. Ambulance or in-catheterization laboratory administration of ticagrelor for primary percutaneous coronary intervention for ST-segment elevation myocardial infarction: rationale and design of the randomized, double-blind Administration of Ticagrelor in the cath Lab or in the Ambulance for New ST elevation myocardial Infarction to open the Coronary artery (ATLANTIC) study. Am Heart J 2013;165:515-22.
- Steinhubl SR, Berger PB, Mann JT III, et al. Early and sustained dual oral antiplatelet therapy following percutaneous coronary intervention: a randomized controlled trial. JAMA 2002;288:2411-20.
- Montalescot G, Bolognese L, Dudek D, et al. Pretreatment with prasugrel in non–ST-segment elevation acute coronary syndromes. N Engl J Med 2013;369:999-1010.
Clinical Topics: Acute Coronary Syndromes, Cardiovascular Care Team, Invasive Cardiovascular Angiography and Intervention, Stable Ischemic Heart Disease, Vascular Medicine, Interventions and ACS, Interventions and Vascular Medicine, Chronic Angina
Keywords: Clopidogrel, Ticagrelor, ST Elevation Myocardial Infarction, Percutaneous Coronary Intervention, Non-ST Elevated Myocardial Infarction, Platelet Membrane Glycoprotein IIb, Acute Coronary Syndrome, Risk, Follow-Up Studies, Hospital Administration, Random Allocation, Aspirin, Thrombosis, Stents, Stroke, Emergency Medical Services, Delivery of Health Care, Randomized Controlled Trials as Topic, Observational Studies as Topic
< Back to Listings