Pericardial Effusion and Postoperative Atrial Fibrillation
Quick Takes
- Postoperative atrial fibrillation (POAF) is the most frequent complication of cardiac surgery patients and is associated with higher length of stay.
- Pericardial effusion plays a pivotal role in the pathogenesis of POAF.
- Posterior pericardiotomy at time of index operation may prevent pericardial effusion and thus POAF.
Postoperative Atrial Fibrillation: What It Is and Why It Matters
Postoperative atrial fibrillation (POAF) has an estimated incidence of 19-30%,1 making it the most frequent complication in cardiac surgery patients.2 At the systems level, POAF is associated with prolonged hospitalization and increased hospital costs.1 At the patient level, POAF is associated with major adverse outcomes including higher rates of postoperative mortality, stroke, heart failure, and renal failure.1,3
The term POAF generally refers to patients with new-onset atrial fibrillation (AF) occurring during the postoperative hospitalization. However, different expert societies have varying criteria for POAF, which may lead to over- or under-diagnosis, mismanagement, and subsequent increased morbidity.4
The most likely timing of onset of POAF is within 48 hours postoperatively, although it can occur anytime within 7 days of surgery.5 The risk of recurrence after leaving the hospital has been reported to be as high as 28% in the first month post-discharge.6
Accepted Mechanisms of POAF
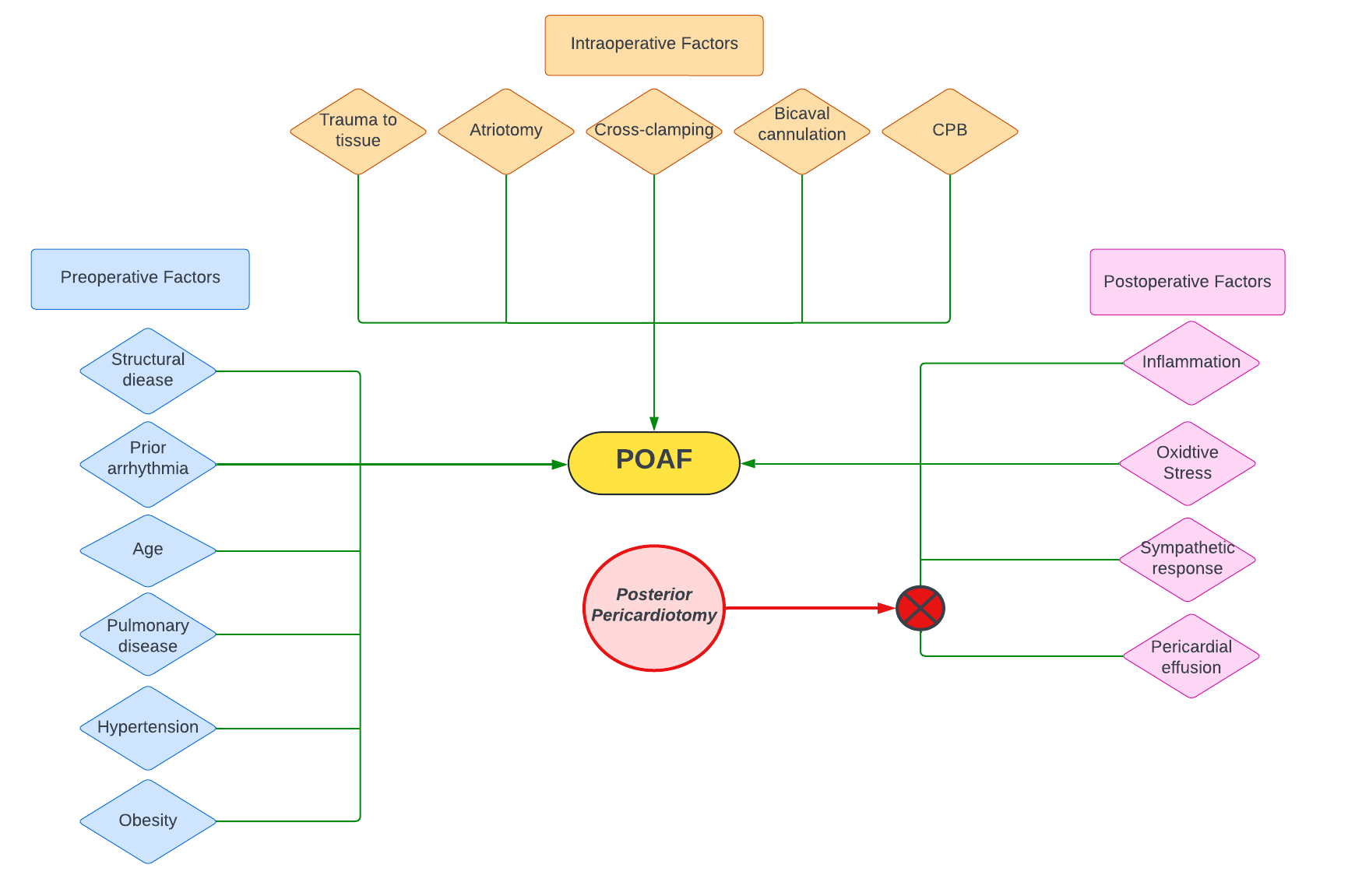
Despite being a postoperative phenomenon, predisposition to developing POAF is present pre-operatively.7 Patients with structural heart disease may be more susceptible to developing POAF, as changes secondary to valvular disease may create re-entry circuits that trigger arrhythmia.8 Fibrosis or fatty infiltration of myocytes, or connexin remodeling with gap junctional modification, can lead to conduction abnormalities. In addition, chronic stress on the atrial substrate caused by numerous other conditions is associated with POAF (Figure 1).5
Figure 1
Intraoperatively, surgical trauma caused by direct injury to the heart or cardiopulmonary bypass (CPB) may lead to remodeling of the atria.9 Bicaval cannulation, duration of CPB, and left ventricular venting via the pulmonary veins have all been associated with a higher risk of POAF (Figure 1).5
The postoperative inflammatory response can lead to aberrant conduction, re-entry arrhythmias, and POAF.10,11 Prior studies have demonstrated a correlation between peak levels of postoperative inflammatory markers and the onset of POAF.12 Corticosteroids have been used in an attempt to reduce the incidence of POAF with inconclusive results. Elevated sympathetic tone may lead to increased heart rate and triggered atrial activity; correspondingly, perioperative beta blockade as well as class III antiarrhythmic use have been demonstrated to result in a lower incidence of POAF (Figure 1).13,14
Pericardial Effusion: Role in POAF
Despite the aforementioned medical management, the overall incidence of POAF has remained high.5 This has generated a hunt for other treatable etiopathologies of POAF.
In 1988 a prospective study of 150 patients undergoing cardiac surgery noted a significantly higher incidence of postoperative supraventricular arrhythmias among patients who developed postoperative pericardial effusion (incidence of POAF: 27% in patients with effusion vs. 11% in patients without effusion, p<0.05).15 Subsequent studies have reported a reduction in POAF with postoperative drainage of the pericardium, suggesting a relationship between pericardial effusion and POAF.16
The presence of mediastinal fluid, regardless of the amount, activates platelets and the coagulation cascade, propagating the inflammatory response via neutrophil migration and increased cytokine release (IL-6, IL-8, IL-1).7,17 As discussed above, surgical trauma itself contributes to inflammation and neutrophil recruitment as well.
Neutrophils produce peroxide, which creates reactive oxygen species; reactive oxygen species are also produced secondary to ischemia-reperfusion injury. The oxidative stress pathway ends with lipid peroxidation of the pericardial fat pads and the atria, engendering POAF.18 Pericardial blood clot hemolysis yields cell-free hemoglobin, which is oxidized to methemoglobin; in turn, methemoglobin reacts with peroxide from neutrophils, which is another path to lipid peroxidation.18,19 Overall, the combination of post-surgical blood products, pericardial and atrial trauma, and oxidative stress secondary to pericardial effusion create a potent pro-inflammatory, pro-arrhythmogenic milieu around the heart that may promote POAF.
Intraoperative Drainage of the Pericardium: A Pre-Emptive Strike
The link between pericardial effusion and POAF is strengthened by studies that have shown a decrease in POAF with pericardial drainage. A propensity-matched analysis of 697 cardiac surgery patients demonstrated that active clearance of pericardial blood products led to significantly lower rates of POAF,20 while another study showed that impaired pericardial drainage was associated with a significantly higher incidence of POAF.21
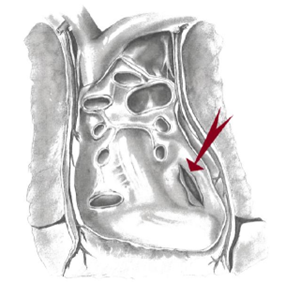
Left posterior pericardiotomy was described by Mulay et al.22 and is a straightforward procedure that creates a 4-5cm longitudinal incision of the posterior pericardium to facilitate drainage of blood products into the left pleural space (Figure 2).
Figure 2
Prior small studies have demonstrated a reduction in POAF among patients receiving posterior pericardiotomy.16 Our group has recently performed a rigorous, adequately powered, randomized controlled trial of posterior pericardiotomy in 420 patients undergoing cardiac surgery and found that posterior pericardiotomy at time of index operation reduced the incidence of pericardial effusion (12% control group vs. 21% pericardiotomy group, respiratory rate [RR] 0.58, confidence interval [CI] 0.37-0.91) and significantly reduced the incidence of POAF (17% in intervention group, 32% in control, p <0.001).23 These results suggest that pericardial effusion plays a potent role in the pathogenesis of POAF, and that improved intraoperative drainage of the pericardium may reduce the incidence of POAF in cardiac surgery patients.
Future Directions
Posterior pericardiotomy is a straightforward procedure that can be performed at time of the index cardiac surgery and may prevent POAF and its negative sequelae (see Figure 1 for graphical summary). However, our trial, while rigorous, was a single-center study. A multi-center study with numerous operators and larger number of subjects will be needed to assure both the efficacy of posterior pericardiotomy and its overall safety prior to its wide adoption as a prophylactic measure against POAF.
References
- Gillinov AM, Bagiella E, Moskowitz AJ, et al. Rate control versus rhythm control for atrial fibrillation after cardiac surgery. N Engl J Med 2016;374:1911–21.
- Lubitz SA, Yin X, Rienstra M, et al. Long-term outcomes of secondary atrial fibrillation in the community: the Framingham Heart Study. Circulation 2015;131:1648–55.
- Eikelboom R, Sanjanwala R, Le ML, Yamashita MH, Arora RC. Postoperative atrial fibrillation after cardiac surgery: a systematic review and meta-analysis. Ann Thorac Surg 2021;111:544–54.
- Filardo G, Pollock BD, da Graca B, et al. Underestimation of the incidence of new-onset post–coronary artery bypass grafting atrial fibrillation and its impact on 30-day mortality. J Thorac Cardiovasc Surg 2017;154:1260–66.
- Rezaei Y, Peighambari MM, Naghshbandi S, et al. Postoperative atrial fibrillation following cardiac surgery: from pathogenesis to potential therapies. Am J Cardiovasc Drugs 2020;20:19–49.
- Lowres N, Mulcahy G, Jin K, Gallagher R, Neubeck L, Freedman B. Incidence of postoperative atrial fibrillation recurrence in patients discharged in sinus rhythm after cardiac surgery: a systematic review and meta-analysis. Interact Cardiovasc Thorac Surg 2018;26:504–11.
- Dobrev D, Aguilar M, Heijman J, Guichard JB, Nattel S. Postoperative atrial fibrillation: mechanisms, manifestations and management. Nat Rev Cardiol 2019;16:417–36.
- Andrade J, Khairy P, Dobrev D, Nattel S. The clinical profile and pathophysiology of atrial fibrillation: relationships among clinical features, epidemiology, and mechanisms. Circ Res 2014;114:1453–68.
- Ishii Y, Schuessler RB, Gaynor SL, et al. Inflammation of atrium after cardiac surgery is associated with inhomogeneity of atrial conduction and atrial fibrillation. Circulation 2005;111:2881–8.
- Ryu K, Li L, Khrestian CM, et al. Effects of sterile pericarditis on connexins 40 and 43 in the atria: correlation with abnormal conduction and atrial arrhythmias. Am J Physiol Heart Circ Physiol 2007;293:H1231–41.
- Zakkar M, Ascione R, James AF, Angelini GD, Suleiman MS. Inflammation, oxidative stress and postoperative atrial fibrillation in cardiac surgery. Pharmacol Ther 2015;154:13–20.
- Lamm G, Auer J, Weber T, Berent R, Ng C, Eber B. Postoperative white blood cell count predicts atrial fibrillation after cardiac surgery. J Cardiothorac Vasc Anesth 2006;20:51–6.
- Echahidi N, Pibarot P, O'Hara G, Mathieu P. Mechanisms, prevention, and treatment of atrial fibrillation after cardiac surgery. J Am Coll Cardiol 2008;51:793–801.
- Arsenault KA, Yusuf AM, Crystal E, et al. Interventions for preventing post-operative atrial fibrillation in patients undergoing heart surgery. Cochrane Database Syst Rev 2013;2013:CD003611.
- Ikäheimo MJ, Huikuri HV, Airaksinen KE, et al. Pericardial effusion after cardiac surgery: incidence, relation to the type of surgery, antithrombotic therapy, and early coronary bypass graft patency. Am Heart J 1988;116:97–102.
- Gozdek M, Pawliszak W, Hagner W, et al. Systematic review and meta-analysis of randomized controlled trials assessing safety and efficacy of posterior pericardial drainage in patients undergoing heart surgery. J Thorac Cardiovasc Surg 2017;153:865-875.e12.
- Wu JHY, Marchioli R, Silletta MG, et al. Oxidative stress biomarkers and incidence of postoperative atrial fibrillation in the omega‐3 fatty acids for prevention of postoperative atrial fibrillation (OPERA) trial. J Am Heart Assoc 2015;4:e001886.
- Youn JY, Zhang J, Zhang Y, et al. Oxidative stress in atrial fibrillation: an emerging role of NADPH oxidase. J Mol Cell Cardiol 2013;62:72–9.
- Kramer PA, Chacko BK, Ravi S, et al. Hemoglobin-associated oxidative stress in the pericardial compartment of postoperative cardiac surgery patients. Lab Invest 2015;95:132–41.
- Baribeau Y, Westbrook B, Baribeau Y, Maltais S, Boyle EM, Perrault LP. Active clearance of chest tubes is associated with reduced postoperative complications and costs after cardiac surgery: a propensity matched analysis. J Cardiothorac Surg 2019;14:192.
- Karimov JH, Gillinov AM, Schenck L, et al. Incidence of chest tube clogging after cardiac surgery: a single-centre prospective observational study. Eur J Cardiothorac Surg 2013;44:1029–36.
- Mulay A, Kirk AJ, Angelini GD, Wisheart JD, Hutter JA. Posterior pericardiotomy reduces the incidence of supra-ventricular arrhythmias following coronary artery bypass surgery. Eur J Cardiothorac Surg 1995;9:150–2.
- Gaudino M, Sanna T, Ballman KV, et al. Posterior left pericardiotomy for the prevention of atrial fibrillation after cardiac surgery: an adaptive, single-centre, single-blind, randomised, controlled trial. Lancet 2021;398:2075–83.
Clinical Topics: Arrhythmias and Clinical EP, Cardiac Surgery, Heart Failure and Cardiomyopathies, Invasive Cardiovascular Angiography and Intervention, Pericardial Disease, Prevention, Valvular Heart Disease, Atrial Fibrillation/Supraventricular Arrhythmias, Cardiac Surgery and Arrhythmias, Cardiac Surgery and Heart Failure, Cardiac Surgery and VHD, Acute Heart Failure, Heart Failure and Cardiac Biomarkers, Interventions and Structural Heart Disease, Interventions and Vascular Medicine, Stress
Keywords: Pericardiectomy, Pericardial Effusion, Prospective Studies, Interleukin-6, Interleukin-8, Methemoglobin, Neutrophils, Reactive Oxygen Species, Atrial Fibrillation, Patient Discharge, Aftercare, Blood Platelets, Cardiopulmonary Bypass, Control Groups, Heart Rate, Hemolysis, Hospital Costs, Lipid Peroxidation, Neutrophil Infiltration, Pulmonary Veins, Respiratory Rate, Pericardium, Reperfusion Injury, Oxidative Stress, Heart Valve Diseases, Adrenal Cortex Hormones, Renal Insufficiency, Heart Failure, Inflammation, Catheterization, Thrombosis, Connexins, Peroxides, Adipose Tissue, Fibrosis, Hospitals, Muscle Cells, Stroke, Interleukin-1
< Back to Listings