Doubt for Vitamin D in Diabetes Prevention
Diabetes mellitus is a major public health problem, with 1.5 million new cases of diabetes diagnosed each year in the United States alone.1 A total of 84 million adults (34% of all adults) in the United States have prediabetes.1 Primary prevention, including dietary approaches, are important in shifting the population distribution to reduce the burden of diabetes. Over the past two decades, vitamin D supplementation has gained popularity as an approach for preventing development of diabetes in individuals with prediabetes, but this concept has not been put to test in large randomized, clinical trials until recently.
The idea behind this concept is rooted in early physiologic studies. The identification of the Vitamin D receptor in the pancreas2 and the finding that reduced insulin secretion in Vitamin D deficiency can be restored by Vitamin D supplementation3 suggested that Vitamin D plays a role in insulin biosynthesis. The upregulation of the insulin receptor in response to vitamin D at insulin-target cells4 and the identification of Vitamin D response elements (VDREs) in the human insulin receptor gene promoter5 suggested that Vitamin D plays a role in insulin uptake.
These physiological findings sparked dozens of prospective studies, the majority of which showed an inverse association between circulating 25(OH)D levels and risk of type 2 diabetes. In a meta-analysis of these prospective studies, each 10 nmol/L increment in 25(OH)D levels was associated with a 4% lower risk of type 2 diabetes.6
Given the promise of these observational findings and the safety, accessibility, and low cost of Vitamin D, numerous randomized, controlled trials (RCTs) studied the effect of Vitamin D supplementation on preventing incident type 2 diabetes. Meta-analysis of these studies showed mixed results.7-9 The largest of these RCTs was the Vitamin D and Type 2 Diabetes (D2d) trial, which were published by Pittas and colleagues in 2019.10
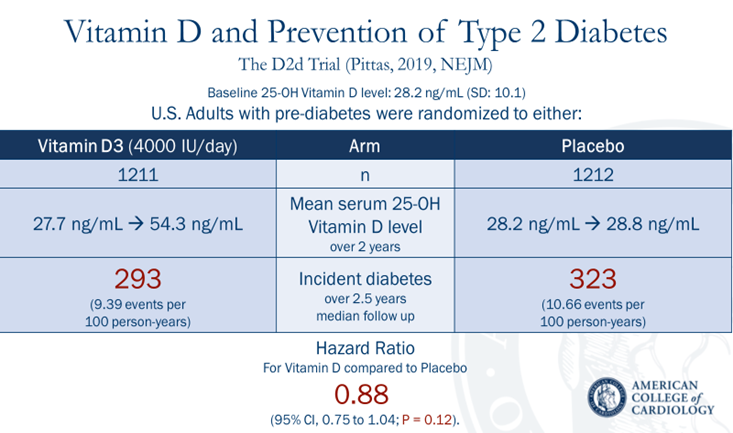
In the D2d trial, 2423 adults with prediabetes across twenty-two medical centers in the United States were randomized to receive 4000 IU per day of Vitamin D3 or to receive placebo, regardless of baseline serum 25-OH Vit D level (Figure 1). After a median follow up of 2.5 years, incident type 2 diabetes occurred in 293 of the 1211 participants in the vitamin D group and 323 of the 1212 participants in the placebo group, corresponding to a hazard ratio (HR) of 0.88 (95% confidence interval 0.75-1.04, p=0.12). While there was no added benefit to vitamin D supplementation in this trial, there was also no added harm to doing so, as the incidence of adverse events, such as hypercalcemia, hypercalciuria, kidney injury, and nephrolithiasis was similar (incidence rate ratio [IRR] 1.23, CI 0.80-1.90).
Figure 1: Visual Abstract of the D2d Trial
D2d was well-designed and used a rigorous protocol, using central laboratory measurement, and assessment of diabetes events in accordance with American Diabetes Association (ADA) guidelines. Other strengths include relative high adherence (86%), low use of off-protocol concomitant therapies, and reduction of confounding by seasonal variability by constant recruiting throughout the calendar year.
However, there are several limitations to the study design which may in part explain the null result. The vast majority (78%) of trial participants already had sufficient levels of vitamin D (≥ 20 ng/mL) at the start of the trial, questioning the utility of supplementation in already-sufficient patients. In a post-hoc analysis of the 103 participants with deficient vitamin D levels (<12 ng/mL), Vitamin D supplementation was associated with reduced incidence of type 2 diabetes (HR 0.38, CI 0.18-0.80). Therefore, what can be concluded from this trial is that if there is any benefit to vitamin D supplementation for diabetes prevention, it does not pertain to a vitamin D-sufficient population.
Secondly, the D2d trial was powered to detect a 25% effect size. Trials often power for larger effect sizes to reduce recruitment requirements. However, risk reduction of 25% may represent too high a bar for primary prevention studies, where even a 5-10% reduction applied across a large population would have a significant impact. Although the target number of 508 diabetes events was reached (at 616), there was no significant difference between the rate of events in each arm of D2d. Of note, the D2d trial comes on the heels of the Tromsø Vitamin D and T2DM trial involving 511 adults with prediabetes in Norway11 and DPVD trial involving 1256 adults with prediabetes in Japan,12 which similarly found non-significant reductions in diabetes risk in prediabetes patients receiving Vitamin D supplementation compared to placebo (HR 0.90, CI 0.69-1.18; and HR 0.87, CI 0.68-1.09; respectively). Because the Tromsø trial and the DPVD trial were powered to detect an effect size of 30% and 36% respectively, the results of these RCTs do not rule out the possibility that diabetes risk may be reduced in prediabetes patients receiving vitamin D supplementation at a smaller effect size, such as 10-15%.
Observational studies in nutritional epidemiology have been limited by intrinsic epistemological problems including over-interpretation of tiny effects that can vary widely in magnitude and direction according to model specification,13 massive confounding, and difficulty obtaining accurate reports of diet intake,14 leading to a call for prioritization of randomized study designs. The results of these RCTs are a prime example of observational findings that do not hold up in randomized, clinical settings. While the results of the D2d trial and the DPVD trial have yet to be incorporated into a meta-analysis, the magnitude of their effect is unlikely to change the null conclusions of Mirhosseini et al's meta-analysis.7 Therefore, at this time, the available evidence does not suggest that Vitamin D should be prescribed for the prevention of diabetes.
References
- Centers for Disease Control and Prevention. National Diabetes Statistics Report, 2017. (https://www.cdc.gov/diabetes/pdfs/data/statistics/national-diabetes-statisticsreport.pdf).
- Johnson JA, Grande JP, Roche PC, Kumar R. Immunohistochemical localization of the 1,25(OH)2D3 receptor and calbindin D28k in human and rat pancreas. Am J Physiol 1994;267:E356–60.
- Labriji-Mestaghanmi H, Billaudel B, Garnier PE, Malaisse WJ, Sutter BC. Vitamin D and pancreatic islet function. 1. Time course for changes in insulin secretion and content during vitamin D deprivation and repletion. J Endocrinol Invest 1998;11:577–84.
- Dunlop TW, Väisänen S, Frank C, Molnár F, Sinkkonen L, Carlberg C. The human peroxisome proliferator-activated receptor delta gene is a primary target of 1alpha,25-dihydroxyvitamin D3 and its nuclear receptor. J Mol Biol 2005;349:248–60.
- Maestro B, Dávila N, Carranza MC, Calle C. Identification of a vitamin D response element in the human insulin receptor gene promoter. J Steroid Biochem Mol Biol 2003;84:223–30.
- Song Y, Wang L, Pittas AG, et al. Blood 25-hydroxy vitamin D levels and incident type 2 diabetes: a meta-analysis of prospective studies. Diabetes Care 2013;36:1422–28.
- Mirhosseini N, Vatanparast H, Mazidi M, Kimball SM. Vitamin D supplementation, glycemic control, and insulin resistance in prediabetics: a meta-analysis. J Endocr Soc 2018;2:687–709.
- Poolsup N, Suksomboon N, Plordplong N. Effect of vitamin D supplementation on insulin resistance and glycaemic control in prediabetes: a systematic review and meta-analysis. Diabet Med 2016;33;290–99.
- Tang H, Li D, Li Y, Zhang X, Song Y, Li X. Effects of vitamin D supplementation on glucose and insulin homeostasis and incident diabetes among nondiabetic adults: a meta-analysis of randomized controlled trials. Int J Endocrinol 2018; Article ID 7908764.
- Pittas AG, Dawson-Hughes B, Sheehan P, et al. Vitamin D supplementation and prevention of type 2 diabetes. N Engl J Med 2019;381:520-30.
- Jorde R, Sollid ST, Svartberg J, et al. Vitamin D 20,000 IU per week for five years does not prevent progression from prediabetes to diabetes. J Clin Endocrinol Metab 2016;101:1647–55.
- Kawahara T. Eldecalcitol, a vitamin D analog, for diabetes prevention in impaired glucose tolerance (DPVD Study). Diabetes 2018;67:120-LB.
- Patel CJ, Burford B, Ioannidis JPA. Assessment of vibration of effects due to model specification can demonstrate the instability of observational associations. J Clin Epidemiol 2015;68:1046–58.
- Trepanowski JF, Ioannidis JPA. Perspective: limiting dependence on nonrandomized studies and improving randomized trials in human nutrition research: why and how. Adv Nutr 2018;9:367–77.
Keywords: Metabolic Syndrome, Vitamin D, Cholecalciferol, Prediabetic State, Insulin, Receptor, Insulin, Vitamin D Response Element, Diabetes Mellitus, Type 2, Receptors, Calcitriol, Prospective Studies, Confidence Intervals, Hypercalcemia, Up-Regulation, Public Health, Follow-Up Studies, Hypercalciuria, Vitamin D Deficiency, Vitamins, Antigens, CD, Nephrolithiasis, Diet, Primary Prevention, Risk Reduction Behavior
< Back to Listings