Contemporary Use of Methods to Evaluate Obstructive CAD in the Catheterization Laboratory
What Is the Role of Coronary Physiology Now (and Beyond)?
In 2020, the long-awaited ISCHEMIA (International Study of Comparative Health Effectiveness with Medical and Invasive Approaches)1 published its findings. The primary conclusion of ISCHEMIA was that among patients with stable coronary disease and moderate or severe ischemia, an initial invasive strategy, compared with an initial conservative strategy, did not reduce the risk of ischemic cardiovascular events or death from any cause (median follow up of 3.2 years).1 To many in the interventional cardiology world, the results seemed at odds with the expectation (supported by observational data),2,3 that compared with medical therapy, vessel-specific ischemia-driven revascularization would translate into improved patient outcomes. Independent of some of the criticisms of ISCHEMIA (principally that the decision to revascularize was primarily based on the result of only noninvasive ischemia testing), as a community we have to ask ourselves: What is the role of coronary physiology now (and beyond)?
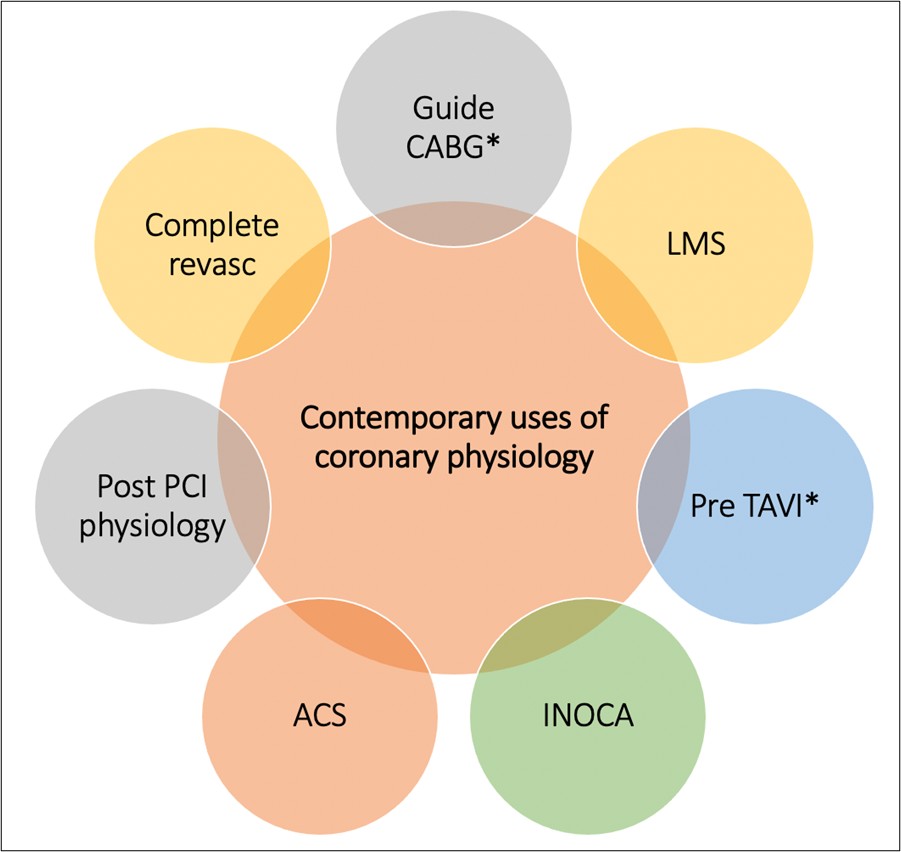
From the early landmark DEFER (Deferral Versus Performance of PTCA in Patients Without Documented Ischemia)4 and FAME (Fractional Flow Reserve Versus Angiography for Multivessel Evaluation)5 studies, coronary physiology has primarily been used for the identification of ischemia to guide revascularization (and its corollary, the deferral of revascularization for stenoses identified as non-flow-limiting). However, post-ISCHEMIA, in certain anatomic subsets where the presence of ischemia per se does not seem to adversely influence patient outcomes, one may argue that the contemporary role of coronary physiology is challenged. Conversely, rather than diminishing in its influence, the role of coronary physiology continues to expand far beyond its initial purpose of determining the functional significance of angiographically intermediate coronary stenoses (Figure 1).
Figure 1: Contemporary Applications of Invasive Coronary Physiology
CABG = coronary artery bypass grafting; LMS = left main stem; TAVI = transcatheter aortic valve implantation; ACS = acute coronary syndrome; INOCA = ischemia and nonobstructive coronary artery disease; PCI = percutaneous coronary intervention
Now more than ever, coronary physiology (both epicardial and microcirculatory) continues to provide the tools to accurately diagnose, guide, and effectively treat patients in the catheterization laboratory. In this brief review, we focus on some of the newer applications of coronary physiology that constitute the contemporary evaluation of obstructive coronary artery disease (CAD) in the catheterization laboratory.
Going Beyond Moderate Lesions: Physiology as Part of State-of-the-Art PCI Practice
Currently, invasive coronary physiology (specifically fractional flow reserve [FFR] and instantaneous wave-free ratio [iFR]) receives a Class IA recommendation to assess the hemodynamic significance of angiographically moderate stenoses.6 However, the contemporary use of physiology has evolved in tandem with the trend of increasing complexity of interventional procedures. Accordingly, the sole purpose of physiology to answer the simple binary question of whether to stent or defer is now outdated. Instead, coronary physiology now forms an integral component of "state-of-the-art" percutaneous coronary revascularization practice.
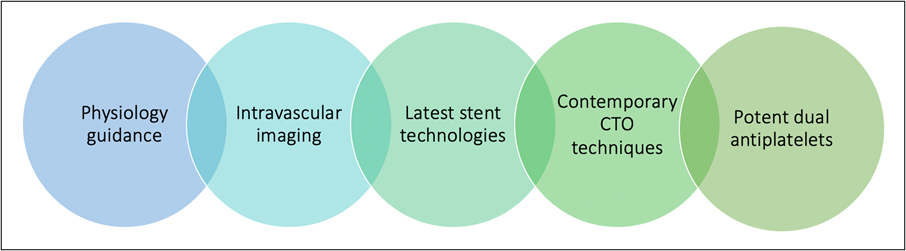
In patients with complex multivessel disease, coronary physiology (particularly by means of the pressure-wire pullback trace) is used not necessarily to adjudicate the need for PCI but rather to guide the stenting strategy to achieve the optimal hemodynamic result. This approach is borne out of the landmark SYNTAX II (State-of-the-Art Percutaneous Coronary Revascularization in de Novo 3 Vessel Disease) trial7, in whom patients with three-vessel CAD at equipoise for CABG (including those with intermediate or high anatomical complexity) underwent physiology-guided revascularization with thin-strut drug-eluting stents, liberal use of intravascular imaging, chronic total occlusion (CTO) techniques, and potent dual antiplatelet therapy (Figure 2).
Figure 2: Schematic Representation of the Key Components of State-of-the-Art PCI Practice
Adopting this approach, PCI undertaken using the SYNTAX II strategy was associated with superior outcomes compared with the PCI arm of the original SYNTAX (Synergy Between Percutaneous Coronary Intervention With TAXUS and Cardiac Surgery) trial and was associated with similar clinical outcomes to the equipoise-derived SYNTAX CABG cohort. Critically, from a physiology viewpoint, technical success of invasive physiological assessment was achieved in >75% of these complex coronary anatomies (3.5 lesions/patient), with only a 0.002% complication rate.7
The Contemporary Coronary Physiology Workflow
As outlined, coronary physiology now adopts a central role in guiding stenting strategies in more complex coronary anatomies. This is principally achieved by the use of non-hyperemic pressure wire pullback curves and, ideally, their co-registration with the coronary angiogram. With non-hyperemic pressure ratio indices, these data allow operators to perform virtual PCI, in which different potential stenting strategies can be modelled to determine the optimal hemodynamic outcome. With the iFR platform, this virtual PCI approach has a demonstrated high degree of accuracy for predicting the final post-PCI result and is associated a significant reduction in stent length and number compared with "spot" physiology measurements.8
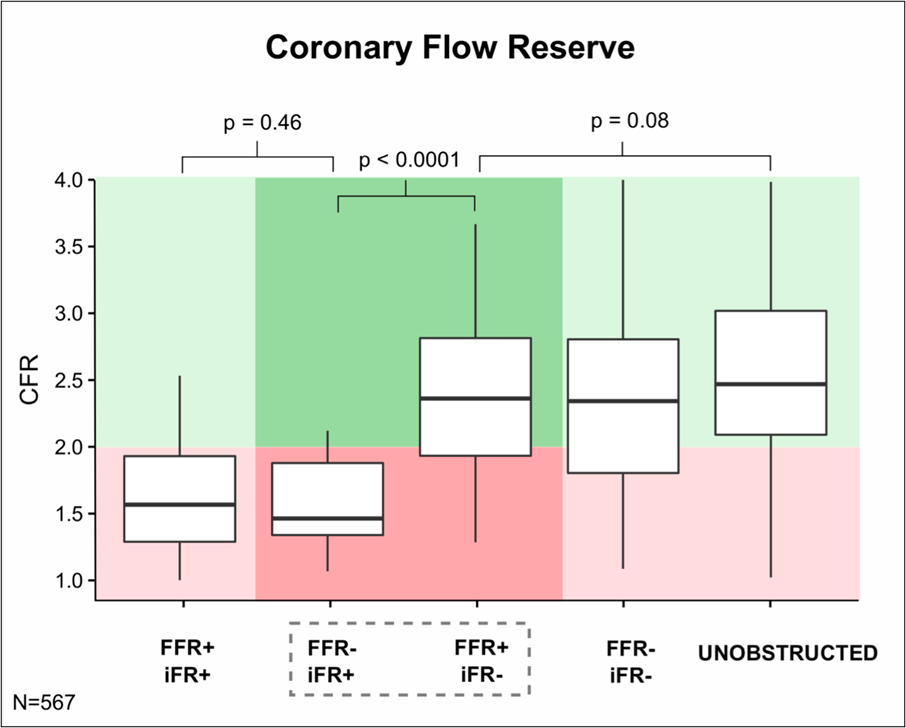
This has led to the development of a contemporary physiology workflow that can be readily incorporated into any PCI procedure (Figure 3). Note that the workflow utilizes both hyperemic and non-hyperemic indices. This is because the combined use of both hyperemic (FFR) and non-hyperemic (iFR) indices can be used to estimate if the underlying coronary flow reserve (CFR) is normal (>2) or abnormal (<2) (Figure 4).9 Lastly, note the integration of post-PCI physiology in the contemporary physiology workflow. Indeed, post-PCI physiology is emerging as an essential application of coronary physiology in modern PCI practice that is backed by an increasingly strong evidence base.
Figure 3: Proposed Contemporary Epicardial Physiology Workflow for Use in Obstructive CAD
Figure 4: Boxplot Comparisons of CFR
The Importance of Post-PCI Coronary Physiology
The 2019 DEFINE PCI study highlighted the importance of systematic measurement of post PCI physiology. In 562 vessels (500 patients) in whom a blinded iFR pull back was performed after operator-assessed angiographically successful PCI, the rate of residual ischemia (iFR <0.90) was assessed.10 The authors detected residual ischemia in nearly 1 in 4 patients after coronary stenting despite an operator-determined angiographically successful result. Furthermore, most cases of residual ischemia were due to inapparent focal lesions potentially amenable to treatment with additional PCI.
The 1-year clinical outcomes of the DEFINE PCI (Blinded Physiological Assessment of Residual Ischemia after Successful Angiographic PCI) study have recently been presented at TCT Connect 2020. The findings indicated that patients who achieved a post-PCI iFR of ≥0.95 had 68% fewer clinical events than patients with less optimal post-PCI iFR values (1.8% vs. 5.7%; p = 0.04).11 These data complement existing post-PCI FFR datasets12 and support "the higher the better" hypothesis regarding obtaining an optimal post-PCI physiological (as opposed to just angiographic) result.
New Frontiers for Coronary Physiology
To conclude, we look briefly at new frontiers for coronary physiology. Regarding the assessment of obstructive CAD, the integration of angiographic, intracoronary imaging and physiologic data ("tri-registration") represents perhaps the most comprehensive strategy to evaluate, plan, and perform modern PCI. Platforms that offer this fully integrated package are likely to become commonplace tools in the contemporary management of obstructive CAD in the catheterization laboratory. There has also been significant interest and development into in the utility of three-dimensional angiography-derived physiological measurements that do not require the use of a pressure guidewire (e.g., quantitative flow ratio).13 Lastly, in the age of artificial intelligence, computational and machine learning algorithms offer significant promise in supporting operators by intelligent interpretation of physiological data14 for optimal PCI outcomes.
Although beyond the scope of this summary, there have also been significant developments in the field of invasive coronary microvascular dysfunction testing in angiographically unobstructed CAD.15 Here coronary physiology plays an essential role in diagnosing the two main pathophysiological mechanisms that can cause coronary microvascular dysfunction: impaired microcirculatory conductance and arteriolar dysregulation. The ability to comprehensively characterize both epicardial and microcirculatory function (by either thermo-dilution, Doppler-wire technologies, and/or the emerging tool of absolute coronary flow)16 is likely to become increasingly commonplace in the catheterization laboratory of the near future.17
References
- Maron DJ, Hochman JS, Reynolds HR, et al. Initial Invasive or Conservative Strategy for Stable Coronary Disease. N Engl J Med 2020;382:1395-407.
- Iskander S, Iskandrian AE. Risk assessment using single-photon emission computed tomographic technetium-99m sestamibi imaging. J Am Coll Cardiol 1998;32:57-62.
- Ladenheim ML, Pollock BH, Rozanski A, et al. Extent and severity of myocardial hypoperfusion as predictors of prognosis in patients with suspected coronary artery disease. J Am Coll Cardiol 1986;7:464-71.
- Bech GJ, De Bruyne B, Pijls NH, et al. Fractional flow reserve to determine the appropriateness of angioplasty in moderate coronary stenosis: a randomized trial. Circulation 2001;103:2928-34.
- Tonino PAL, De Bruyne B, Pijls NHJ, et al. Fractional flow reserve versus angiography for guiding percutaneous coronary intervention. N Engl J Med 2009;360:213-24.
- Knuuti J, Wijns W, Saraste A, et al. 2019 ESC Guidelines for the diagnosis and management of chronic coronary syndromes. Eur Heart J 2020;41:407-77.
- Escaned J, Collet C, Ryan N, et al. Clinical outcomes of state-of-the-art percutaneous coronary revascularization in patients with de novo three vessel disease: 1-year results of the SYNTAX II study. Eur Heart J 2017;38:3124-34.
- Kikuta Y, Cook CM, Sharp ASP, et al. Pre-Angioplasty Instantaneous Wave-Free Ratio Pullback Predicts Hemodynamic Outcome In Humans With Coronary Artery Disease: Primary Results of the International Multicenter iFR GRADIENT Registry. JACC Cardiovasc Interv 2018;11:757-67.
- Cook CM, Jeremias A, Petraco R, et al. Fractional Flow Reserve/Instantaneous Wave-Free Ratio Discordance in Angiographically Intermediate Coronary Stenoses: An Analysis Using Doppler-Derived Coronary Flow Measurements. JACC Cardiovasc Interv 2017;10:2514-24.
- Jeremias A, Davies JE, Maehara A, et al. Blinded Physiological Assessment of Residual Ischemia After Successful Angiographic Percutaneous Coronary Intervention: The DEFINE PCI Study. JACC Cardiovasc Interv 2019;12:1991-2001.
- Patel M, Jeremias A, Davies J, et al. One-year outcomes of patients with residual physiologic ischemia after percutaneous coronary intervention: the DEFINE PCI trial. Presented at: TCT 2020. October 15, 2020.
- Rimac G, Fearon WF, De Bruyne B, et al. Clinical value of post-percutaneous coronary intervention fractional flow reserve value: A systematic review and meta-analysis. Am Heart J 2017;183:1-9.
- Collet C, Onuma Y, Sonck J, et al. Diagnostic performance of angiography-derived fractional flow reserve: a systematic review and Bayesian meta-analysis. Eur Heart J 2018;39:3314-21.
- Cook CM, Takayuki W, Howard JP, et al. Algorithmic Versus Expert Human Interpretation of Instantaneous Wave-Free Ratio Coronary Pressure-Wire Pull Back Data. JACC Cardiovasc Interv 2019;12:1315-24.
- Ford TJ, Stanley B, Good R, et al. Stratified Medical Therapy Using Invasive Coronary Function Testing in Angina: The CorMicA Trial. J Am Coll Cardiol 2018;72:2841-55.
- Xaplanteris P, Fournier S, Keulards DCJ, et al. Catheter-Based Measurements of Absolute Coronary Blood Flow and Microvascular Resistance: Feasibility, Safety, and Reproducibility in Humans. Circ Cardiovasc Interv 2018;11:e006194.
- Kunadian V, Chieffo A, Camici PG, et al. An EAPCI Expert Consensus Document on Ischaemia with Non-Obstructive Coronary Arteries in Collaboration with European Society of Cardiology Working Group on Coronary Pathophysiology & Microcirculation Endorsed by Coronary Vasomotor Disorders International Study Group. Eur Heart J 2020;41:3504-20.
Clinical Topics: Acute Coronary Syndromes, Cardiac Surgery, Invasive Cardiovascular Angiography and Intervention, Stable Ischemic Heart Disease, Atherosclerotic Disease (CAD/PAD), Aortic Surgery, Cardiac Surgery and SIHD, Interventions and ACS, Interventions and Coronary Artery Disease, Chronic Angina
Keywords: Angina, Stable, Fractional Flow Reserve, Myocardial, Drug-Eluting Stents, Taxus, Platelet Aggregation Inhibitors, Coronary Artery Disease, Percutaneous Coronary Intervention, Acute Coronary Syndrome, Artificial Intelligence, Constriction, Pathologic, Transcatheter Aortic Valve Replacement, Laboratories, Microcirculation, African Americans, Vibrissae, Introduced Species, Follow-Up Studies, Motivation, Coronary Stenosis, Coronary Artery Bypass, Ischemia, Hemodynamics, Catheterization
< Back to Listings