Integrating CAC Scores Into Risk Stratification of Patients Undergoing Cancer Treatment
Quick Takes
- Automated coronary artery calcium (CAC) scores can serve as accurate predictors of adverse cardiac outcomes among oncologic patients undergoing chemoradiotherapy.
- CAC scores calculated on routine nongated chest CT should be used to risk-stratify oncologic patients before treatment and may help modulate the cardiotoxic effects of cancer therapies through the addition of cardioprotective regimens for patients at high risk.
- Pretreatment CAC scores can be applied to the sample selection for randomized controlled trials evaluating novel cardioprotective therapies.
Introduction
Recent advances in cancer treatment have increased survival rates. Survivors, however, often face significant morbidity and mortality from the adverse cardiac effects of chemoradiation, both during and after completion of treatment. Anthracycline and trastuzumab chemotherapeutics, for example, have been associated with left ventricular systolic dysfunction and heart failure.1 Radiation therapy is linked with an increased risk of atherosclerosis, an increased rate of progression of atherosclerosis, and acute coronary events.2
Risk Assessment Technologies
Historically, prediction of cancer therapy-related cardiac dysfunction (CTRCD) outcomes and other adverse cardiovascular disease (CVD) outcomes has been based on the presence of baseline traditional CVD risk factors and echocardiographic findings. Although patients with existing CVD risk factors are at higher risk of CTRCD, there is significant heterogeneity in observed CVD outcomes. Cardiac magnetic resonance imaging can be used to evaluate cardiomyopathy through the characterization of ventricular size and function but is costly and time-consuming. Breast arterial calcification visualized on mammography is associated with coronary artery calcium (CAC) and an increased risk of CVD,3 although the optimal strategy for quantifying and reporting breast arterial calcification is uncertain.
Electrocardiography (ECG)-gated noncontrast cardiac computed tomography (CT) is a widely available tool that quantifies an individual's burden of CAC. Higher CAC scores are strongly associated with endothelial dysfunction and increased CVD risk and are a marker of overall suboptimal health. This scoring is advantageous for patients with many types of cancer because nongated chest CT is routinely performed as part of the planning for chemoradiation, and CAC scoring obtained from these noncardiac CT chests provide very similar results to formal ECG-gated CAC scans. Furthermore, artificial intelligence and deep-learning algorithms have fostered the development of automated CAC scoring, thereby reducing another barrier to the reporting and use of CAC scores. Importantly, these automated CAC scores are accurate, offering a noninvasive, fast, simple, and economic tool to calculate CAC scores using standard nongated chest CTs.4
Implications of CAC Scores Among Oncologic Patients Before Treatment
Pretreatment CAC score >0 has been associated with an increased risk of adverse cardiac outcomes among patients undergoing radiation and post chemotherapy for patients with breast or lung cancer. Patients with breast or lung cancer with pretreatment CAC score >0 experience poorer cardiac outcomes, particularly those that are vascular in nature.2,5 Higher doses of radiation therapy and anthracycline-based chemotherapies have been associated with even worse outcomes.2,6 However, whether CAC score >0 improved risk stratification for patients with diffuse large B-cell lymphoma (DLBCL), who commonly undergo treatment with anthracyclines, was uncertain.
In a retrospective, multicenter study, Shen et al. examined 1,468 patients with DLBCL who underwent a nongated chest CT before initiation of anthracycline-based chemotherapy. CAC score 1-100 was associated with 2.6 times higher odds of CTRCD (defined as a reduction in left ventricular ejection fraction [LVEF] between 10% and 50%) than the odds for those with CAC score of 0, and CAC score >100 with 5.2 times higher odds of CTRCD and 3.7 times higher risk of a major adverse cardiac event (MACE) for approximately 6 years following the scoring. The risk of MACE was 3.7 times higher among those with CAC score 1-100 and 7.9 times higher among those with CAC score >100 than for those with CAC score of 0,6 which may be due to anthracycline-induced microcirculation damage within the coronary arteries.7 These findings demonstrate that patients with baseline CAC score >0, as determined on a nongated chest CT, should be considered for an individualized cancer therapy regimen that includes concomitant cardioprotective agents together with careful consideration of the risks and benefits of their chosen chemoradiation regimen.
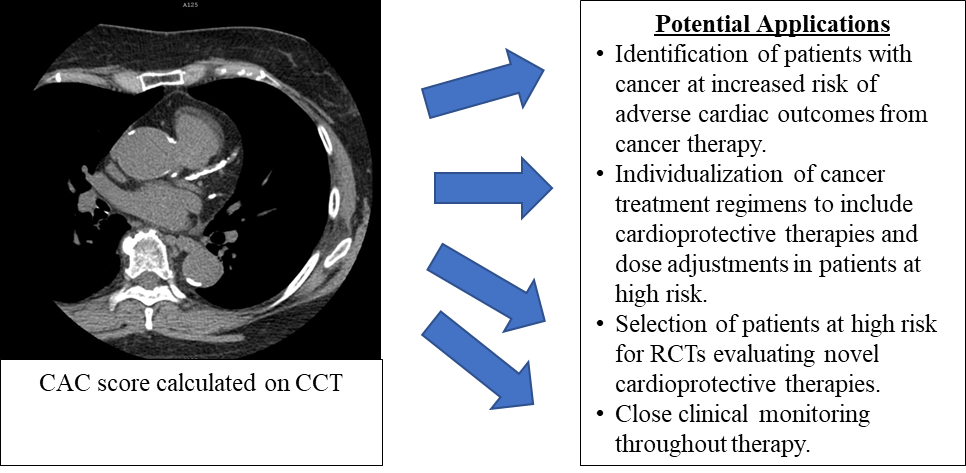
Given the potential implications of pretreatment CAC scores among oncologic patients (Figure 1), some professional societies now recommend their implementation in cancer therapy planning. A 2022 expert consensus document from the Society of Cardiovascular Computed Tomography (SCCT) provided a strong recommendation for the calculation of a CAC score for all oncologic patients using their routine nongated chest CTs before initiation of therapy to better risk-stratify patients and incorporate appropriate preventive therapies in their cancer treatments.8
Figure 1: Applications of CAC Scores in Cancer Treatment
Historically, the presence of CV risk factors and echocardiographic biomarkers have been relied on to identify oncologic patients at high risk of adverse cardiac outcomes. Imaging provides a low-cost, simple, fast, accurate, and reliable tool for the identification of patients at high risk who may benefit from: 1) cardioprotective therapies in conjunction with their cancer treatments; 2) RCTs investigating cardioprotective therapies for patients with cancer; and 3) close clinical monitoring throughout the duration of treatment.
CAC = coronary artery calcium; CCT = cardiac computed tomography; CV = cardiovascular; RCT = randomized controlled trial.
To balance the risks and benefits of cancer therapeutics, efforts have begun to identify ways to ameliorate the associated cardiotoxicity of many. Neurohormonal antagonists, such as beta-blockers and angiotensin-converting enzyme inhibitors/angiotensin-receptor blockers (ACEIs/ARBs), along with statins, have been shown to attenuate the reduction in LVEF among patients undergoing treatment with anthracycline and/or trastuzumab.9
These studies suggest that the concomitant use of beta-blockers, ACEIs/ARBs, or statins with chemotherapeutics in patients at high risk can both improve the short-term cardiac outcomes and lengthen cardiotoxicity-free time, which may improve adherence to the chemotherapy regimen. Oncologic patients identified as being at high risk can have their treatment regimen further modified by lowering the dose of chemoradiation provided, ameliorating the cardiotoxicity.2,6
Although several cardioprotective therapies have shown promising results regarding the attenuation of a decline in LVEF, important knowledge gaps remain. Future research should investigate the long-term outcomes of cardioprotective measures and the required dose for optimal prevention. A large amount of heterogeneity regarding observed benefit exists within the literature, largely because of differences in study populations and cancers examined. The large heterogeneity has been hypothesized as an explanation for the small level of attenuation in LVEF reduction and the absence of significant improvement in clinical outcomes despite a trend toward lower rates of adverse clinical events.10 The study by Shen et al. provided further support for the use of CAC score quantified from pretreatment nongated chest CTs to identify oncologic patients at higher risk of adverse cardiac outcomes, which may aid in enrollment strategies in the future. Trials are needed to elucidate optimal dosing, timing of cardioprotective treatment initiation, long-term outcomes, and clinical implications of adjuvant cardioprotective agents. The use of CAC score to better identify the patients most likely to benefit from personalized cardioprotective regimens is an important opportunity that may help clarify guideline recommendations.
References
- Lee ARYB, Yau CE, Low CE, et al. Natural progression of left ventricular function following anthracyclines without cardioprotective therapy: a systematic review and meta-analysis. Cancers (Basel) 2023;15:512.
- Kim K, Chung SY, Oh C, et al. Automated coronary artery calcium scoring in patients with breast cancer to assess the risk of heart disease following adjuvant radiation therapy. Breast 2022;65:77-83.
- McLenachan S, Camilleri F, Smith M, Newby DE, Williams MC. Breast arterial calcification on mammography and risk of coronary artery disease: a SCOT-HEART sub-study. Clin Radiol 2019;74:421-8.
- Xu C, Guo H, Xu M, et al. Automatic coronary artery calcium scoring on routine chest computed tomography (CT): comparison of a deep learning algorithm and a dedicated calcium scoring CT. Quant Imaging Med Surg 2022;12:2684-95.
- Koutroumpakis E, Xu T, Lopez-Mattei J, et al. Coronary artery calcium score on standard of care oncologic CT scans for the prediction of adverse cardiovascular events in patients with non-small cell lung cancer treated with concurrent chemoradiotherapy. Front Cardiovasc Med 2022;Dec 2:[ePub ahead of print].
- Shen H, Lian Y, Yin J, et al. Cardiovascular risk stratification by automatic coronary artery calcium scoring on pretreatment chest computed tomography in diffuse large B-cell lymphoma receiving anthracycline-based chemotherapy: a multicenter study. Circ Cardiovasc Imaging 2023;Feb 7:[ePub ahead of print].
- Galán-Arriola C, Vílchez-Tschischke JP, Lobo M, et al. Coronary microcirculation damage in anthracycline cardiotoxicity. Cardiovasc Res 2022;118:531-41.
- Lopez-Mattei J, Yang EH, Baldassarre LA, et al. Cardiac computed tomographic imaging in cardio-oncology: an expert consensus document of the Society of Cardiovascular Computed Tomography (SCCT). Endorsed by the International Cardio-Oncology Society (ICOS). J Cardiovasc Comput Tomogr 2023;17:66-83.
- Lewinter C, Nielsen TH, Edfors LR, et al. A systematic review and meta-analysis of beta-blockers and renin-angiotensin system inhibitors for preventing left ventricular dysfunction due to anthracyclines or trastuzumab in patients with breast cancer. Eur Heart J 2022;43:2562-9.
- Vaduganathan M, Hirji SA, Qamar A, et al. Efficacy of neurohormonal therapies in preventing cardiotoxicity in patients with cancer undergoing chemotherapy. JACC CardioOncol 2019;1:54-65.
Clinical Topics: Noninvasive Imaging, Computed Tomography, Nuclear Imaging, Prevention, Heart Failure and Cardiomyopathies, Cardio-Oncology
Keywords: Tomography, X-Ray Computed, Chemoradiotherapy, Calcium, Atherosclerosis, Cancer
< Back to Listings