Cover Story | The Digital Operating Room: Patient-Specific Modeling For Optimal Structural Heart Outcomes

In a complex TAVR case, the analysis generated from the traditional preprocedural planning seemed reassuring. Annular dimensions appeared within range, coronary heights looked safe and the plan followed standard criteria. Yet when the patient's anatomy was modeled using the DASI Simulations computational modeling platform, a different story emerged. The dynamic interaction between the valve and calcified tissue revealed a previously overlooked risk that static measurements had missed. With this knowledge, the clinical team adapted their strategy and sent this patient to receive a surgical aortic valve replacement (SAVR) instead of a TAVR.
This case illustrates a broader problem: traditional planning tools reduce a living, deformable system to static numbers. The heart, however, is never static. Tissue compliance, calcium distribution and device expansion all interact in ways that imaging snapshots cannot fully capture. Dynamic biomechanical simulation bridges this gap, transforming procedural planning from 2D measurements into a predictive view of real-world device and anatomy interaction.
The Dynamic Difference
Traditional TAVR planning still relies heavily on static CT measurements, including annular diameters, coronary heights and sinus dimensions, which are frozen in 2D slices. These numbers help the team choose a device size, but they don't reveal how a stent frame will actually behave once it meets irregular calcium, compliant tissue and complex root geometry. In other words, a "one-size-fits-all" plan risks overlooking patient-specific realities.
Avoiding complications is challenging enough; however, what is increasingly at stake is the quality of the very first implant. As TAVR expands into younger populations, the initial device must not only function reliably for decades but also preserve options for future interventions. Optimizing that first implant, both for durability and for lifetime management, has become a central focus in the evolution of TAVR planning.
Biomechanical computational models as developed at Georgia Institute of Technology can shift this paradigm. Instead of treating anatomy as a rigid structure, artificial intelligence (AI)-based simulations can model how a valve expands and interacts with surrounding tissue in 3D (Figure 1). This includes predicting how leaflets will displace, how the stent frame may deform and where contact pressures will peak. By capturing these dynamic factors, clinicians gain a preview of deployment that static planning cannot provide.
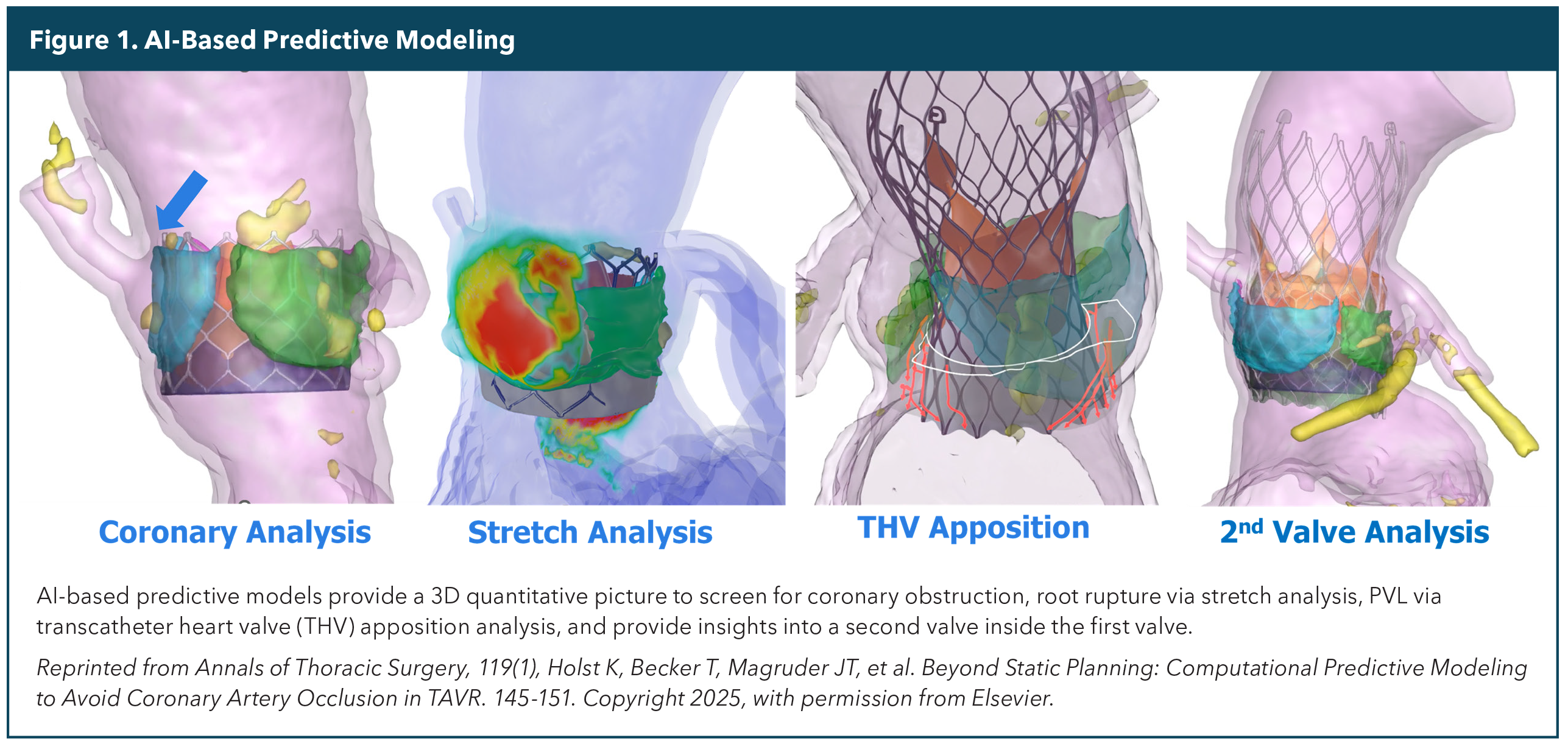

The value becomes clear in high-risk anatomical scenarios, such as bicuspid valves, bulky calcification, low coronary heights or valve-in-valve cases where millimeters matter. In these contexts, AI-based computational modeling offers not just better visualization but actionable insight, helping heart teams anticipate complications and tailor strategies before ever entering the cardiac catheterization lab.
Clinical Applications and Evidence
Over the past decade, TAVR has reshaped the treatment of aortic stenosis. What began as a lifeline for patients with inoperable conditions has now evolved into a frontline therapy, with large randomized clinical trials demonstrating strong results even in low-risk populations. By 2020, more than 400,000 patients worldwide had undergone TAVR, a number that continues to grow as the procedure proves less invasive and more accessible than open surgery.1
But success has not erased risk. Complications such as paravalvular leak (PVL), conduction disturbances requiring pacemaker implantation, valve thrombosis or even aortic root rupture remain concerns. As younger and healthier patients are treated, the stakes rise: these individuals will likely live decades with their prosthetic valves, making durability and complication avoidance paramount. Optimized valve sizing, precise positioning and careful selection are no longer optional; they are essential.
AI-based computational modeling has emerged as a mechanism to assist in refining these decisions. By simulating the actual deployment process, clinicians can better assess the risk of coronary obstruction, anticipate how calcified nodules might redirect frame expansion or determine whether a particular valve size will achieve maximum stent apposition while mitigating aortic root rupture in the sinuses. Studies evaluating these tools have shown encouraging predictive accuracy, suggesting they can help reduce adverse events and improve procedural success rates. The paradigm can be changed from solely identifying the size to personalizing for lifelong durability and complication reduction.
Recent studies highlight how simulations and computational modeling can enhance decision-making in the most complex anatomies. For example, Becker and colleagues evaluated the role of computational predictive modeling in bicuspid aortic valves, where asymmetric leaflets, heavy calcification and elliptical annuli often lead to unpredictable stent expansion. Their work demonstrated that simulation could anticipate frame deformation and leaflet displacement patterns that standard CT planning could not, providing clinicians with a clearer view of how device-tissue interaction might compromise outcomes. Notably, the models highlighted risks of PVL and root injury, two complications that remain especially problematic in bicuspid interventions.2
Similarly, Holst, et al., applied these AI-based predictive simulations to patients at elevated risk of coronary obstruction during TAVR.3 In this prospective series of 116 high-risk patients, computational modeling identified those most likely to encounter compromised coronary flow, guiding modifications such as bioprosthetic or native aortic scallop intentional laceration to prevent iatrogenic coronary artery obstruction (BASILICA), chimney stenting or tailored depth of implant. Among patients predicted to be high risk, none experienced coronary occlusion when strategies were adjusted according to the simulation results (Figure 2).4
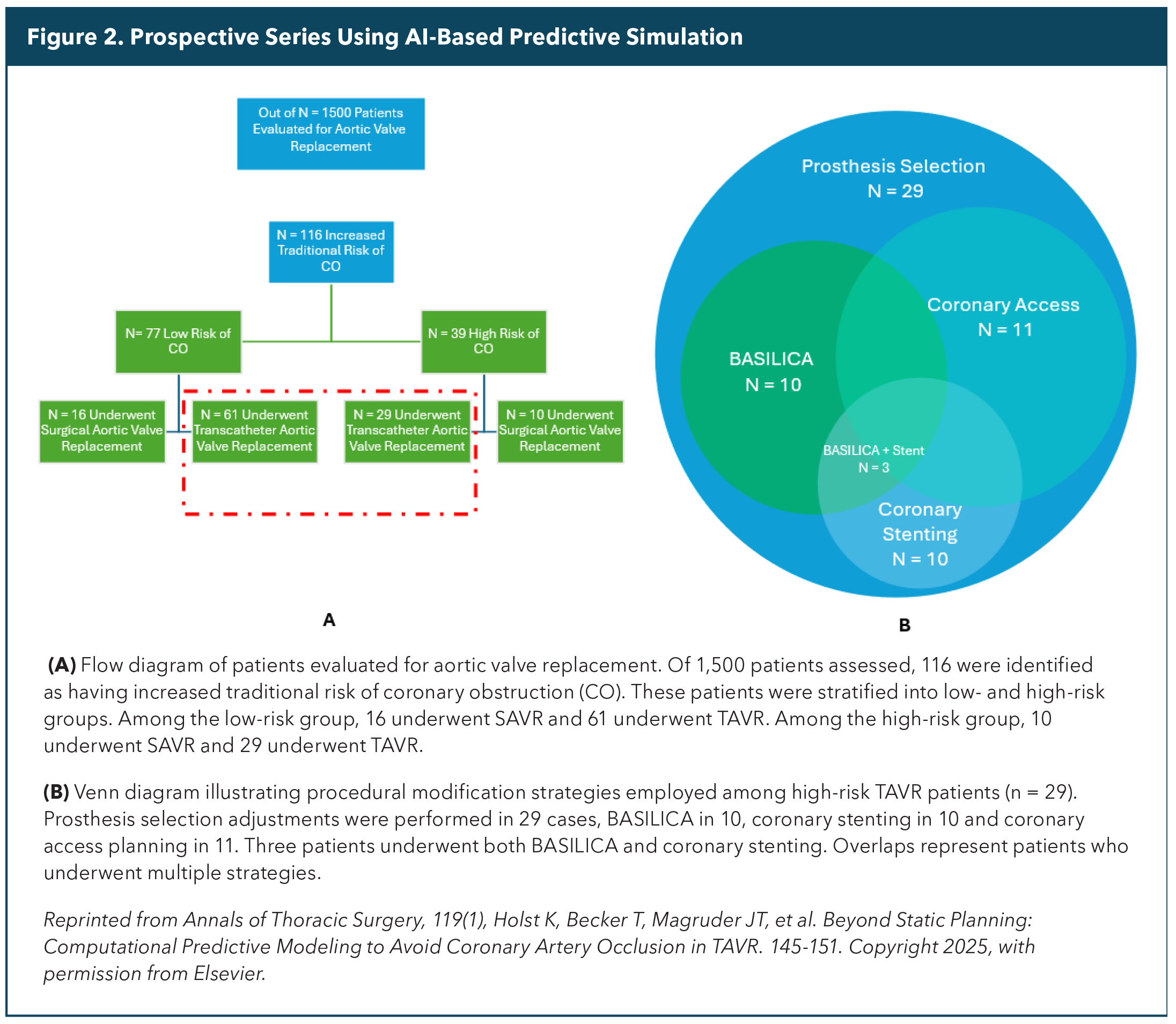
Together, these studies reinforce the central value of computational modeling: not simply visualizing anatomy, but forecasting the dynamic, patient-specific consequences of device deployment. AI models will soon be able to predict post TAVR gradient as well as the risk of thrombosis.5,6 In doing so, simulation moves planning beyond static measurements toward proactive, individualized strategies that help mitigate catastrophic complications.
The need is amplified as TAVR expands into more hospitals and community settings. While access improves, variation in preprocedural planning quality raises concerns about inequities in care. A standardized simulation-driven approach could help ensure consistent planning rigor regardless of geography, supporting heart teams in both academic and regional centers.
Workflow Integration

For new technologies to make a real difference, they must fit seamlessly into existing workflows. Traditional finite element analysis has long been the gold standard in biomechanics. However, its complexity and heavy computational demands make it impractical for the real-time clinical decisions that a multidisciplinary cardiac care team must make.
Newer physics-based particle models, such as the U.S. Food and Drug Administration cleared PrecisionTAVI framework from DASI Simulations, that are now reimbursed by Medicare, are designed to overcome that barrier. These models run in minutes rather than days, while still capturing the essential valve-tissue mechanics that matter for planning. The result is a tool that fits within the typical timeline from CT acquisition to preprocedure heart team conference. Teams can review the simulation results in conjunction with their current preplanning analysis to create a more holistic picture of the patient's unique anatomy.
Practice Implications
Incorporating simulations into preprocedural planning is not meant to replace clinical judgment – it's about enhancing it. By quantifying likely deformation, contact pressures and flow pathways, biomechanical modeling provides the heart team with a richer evidence base for discussion. Communicating these insights to patients can also enhance shared decision-making, as risks and trade-offs become more visible rather than abstract.
For hospitals, reducing complications has both clinical and economic impact. TAVR remains one of the costliest procedures in cardiovascular medicine, with valve prostheses alone accounting for the majority of reimbursement. Each avoided complication not only spares patients from morbidity but also helps institutions optimize resource use in an era of tightening margins.
Future Directions

The future of TAVR planning lies in making simulation smarter and faster. Coupling particle-based biomechanics with machine learning opens the door to instant predictions at the point of care. Beyond preprocedure planning, these models may soon guide operators during the intervention itself, adjusting in real time as deployment unfolds.
More broadly, the trajectory points toward personalization. As medicine shifts from population averages to patient-specific care, dynamic simulation exemplifies the kind of technology that will enable tailored strategies, extending not only to TAVR but to structural heart interventions of all kinds.
This article was authored by Taylor Becker, PhD, Department of Biomedical Sciences, The Ohio State University, Columbus, OH, and Lakshmi Prasad Dasi, PhD, FACC, Department of Biomedical Engineering, Georgia Institute of Technology and Emory University, Atlanta, GA, and founder and chief technology officer of DASI Simulations.
References
- Abugroun A, Daoud H, Abdel-Rahman ME, et al. National trends of outcomes in transcatheter aortic valve replacement (tavr) through transapical versus endovascular approach: From the National Inpatient Sample (NIS). Cardiovasc Revasc Med 2020;21:964-70.
- Becker TS, Holst K, Yadav P, Dasi L, Thourani V. TCT-618 Computational Predictive Modeling for Bicuspid Transcatheter Aortic Valve Replacements. JACC 2023;82:B248.
- Holst K, Becker T, Magruder JT, et al. Beyond static planning: computational predictive modeling to avoid coronary artery occlusion in TAVR. Ann Thorac Surg 2025;119:145-51.
- Becker T, Holst K, Yadav P, et al. Computational predictive modeling for bicuspid transcatheter aortic valve replacements. JACC 2024;83:1116.
- Song W, Sirset-Becker T, Mata Quinonez LR, et al. Machine learning methods to predict transvalvular gradient waveform post–transcatheter aortic valve replacement using preprocedural echocardiogram. J Thorac Cardiovasc Surg 2025;170:1402-1409.e1.
- Venkatesh A, Esmailie F, Tregobov N, et al. A novel computational method to predict hypoattenuated leaflet thickening post-transcatheter aortic valve replacement using preprocedural computed tomography scans. JTCVS Structural Endovascular 2025;5:100041.
Clinical Topics: Cardiac Surgery, Invasive Cardiovascular Angiography and Intervention, Noninvasive Imaging, Interventions and Imaging, Computed Tomography, Nuclear Imaging
Keywords: Cardiology Magazine, ACC Publications, Tomography, X-Ray Computed, Computer Simulation, Transcatheter Aortic Valve Replacement, Artificial Intelligence
