Rationale For the Third Dimension in Preoperative Assessments of Atrioventricular Septal Defects
Quick Takes
- Three-dimensional echocardiography (3DE) adds important information to two-dimensional echocardiography in identifying anatomical abnormalities in the atrioventricular valve (AVV) that contribute to postoperative left atrioventricular valve regurgitation (LAVVR) in atrioventricular septal defect (AVSD).
- Routine quantitative assessment with 3DE of the AVV before surgery may improve prognostication of outcomes and mitigate the risk of severe postoperative LAVVR, highlighting the need for advanced quantitative tools for more accurate valve analysis.
- Despite improvements in surgical techniques for AVSD repair, the persistence of significant postoperative LAVVR in a substantial percentage of patients underscores the importance of further research and innovation in both imaging and surgical care to reduce this common issue.
Although surgical death during atrioventricular septal defect (AVSD) repair has declined in the past decades, significant postoperative left atrioventricular valve regurgitation (LAVVR) remains in up to 25% of patients. A recent, prospective, multicenter observational study had findings of approximately 35% of patients post-AVSD repair having major residual lesions, and 5% of the cohort required reintervention within 30 days, with most reinterventions due to LAVVR.1 Well-established insights from surgical series have identified rare anatomical risk factors, the severity of preoperative LAVVR, and other patient factors for increased risk of postoperative LAVVR (Table 1).2 Protective features include a superior bridging leaflet that spans into the right ventricle (Rastelli type C) and an inferior bridging leaflet without a significant cleft, a valve morphology more commonly associated with trisomy 21. Earlier surgical techniques, such as the single patch repair with division of the bridging leaflets during repair, also has increased risk of postoperative LAVVR and is no longer performed in the modern era. Current repairs are performed with the modified single patch or double patch repair with no difference in outcomes.3
Table 1: Preoperative Risk Factors for Postoperative LAVVR
| Surgical Series and 2DE | 3DEa |
| Greater-than-moderate regurgitation | Qualitative Features |
| Double-orifice LAVV | Commissural abnormalities |
| Single papillary muscle | Asymmetric SBL/IBL leaflet size |
| Dysplastic leaflets | Unbalanced papillary muscle size |
| Younger age at surgery | – |
| Heterotaxy | Quantitative Features |
| – | Reduced (more acute) anterior papillary muscle-to-annulus angle |
| Protective Factors | Shorter APM chords |
| Trisomy 21 | – |
| Free-floating SBL and IBL without cleft |
aIn addition to first column
2DE = two-dimensional echocardiography; 3DE = three-dimensional echocardiography; APM = anterior papillary muscle; IBL = inferior bridging leaflet; LAVV = left atrioventricular valve; LAVVR = left atrioventricular valve regurgitation; SBL = superior bridging leaflet.
Since the introduction of two-dimensional echocardiography (2DE) in the 1970s for assessment of congenital heart defects, there are surprisingly few studies that have extended clinical understanding of the development of postoperative LAVVR. With the introduction of real-time three-dimensional echocardiography (3DE) in 2006, 3DE has identified commissural abnormalities, annular dilation, leaflet prolapse, and reduced (more acute) angles of the subvalvular apparatus to the annular plane in follow-up of patients with postoperative LAVVR.4 Published 3DE case series and surgical series of patients with AVSD repair who had complete closure of the cleft have found anatomical abnormalities in the leaflet commissures, papillary muscle size and position, or chords, or an imbalance of superior and inferior bridging leaflet size, to be risk factors in the development of severe postoperative LAVVR.5,6 These findings challenged the surgical paradigm of complete closure of the cleft in the left atrioventricular valve (LAVV) with two papillary muscles and led to a change in the surgical management of AVSD at the author's institution. As 3DE has been shown to be superior to 2DE in identifying these subtle atrioventricular valve (AVV) abnormalities (Video 1), as well as being more sensitive and specific for AVV regurgitation jet locations, all AVSD now undergo a preoperative 3DE to identify the presence of these features for risk stratification. The perceived risk and benefit of complete, partial, or nonclosure of the LAVV cleft is discussed with the surgeon to avoid early severe LAVVR. Although there was initial concern that this approach might lead to an increased risk of long-term LAVVR, the results of a recent, large Australian cohort of propensity-matched patients with cleft closed versus cleft open included no difference in LAVV reoperations at 15-year follow-up.7
Video 1
Compares the findings on preoperative 2DE of subcostal slow sweep in the en face view of an infant with complete AVSD LAVV, and the findings of the preoperative 3DE of the same patient.
2DE = two-dimensional echocardiography; AO = aortic valve; APM = anterior papillary muscle; AVSD = atrioventricular septal defect; IBL = inferior bridging leaflet; LAVV = left atrioventricular valve; ML = mural leaflet; PPM = posterior papillary muscle; RA = right anterior leaflet; RL = right lateral leaflet; SBL = superior bridging leaflet.
Recently, Nam et al. conducted a 3DE study of 35 patients with complete atrioventricular septal defect (CAVSD) in which they found that patients with postoperative LAVVR had a flatter preoperative annulus at end-systole, whereas leaflet or papillary muscle variables were not associated.8 In contrast, the author's quantitative 3DE analysis on a preoperative cohort of 52 patients with partial AVSD and 77 patients with CAVSD with balanced ventricles and so-called normal LAVV (two papillary muscles and no leaflet dysplasia or double orifice) found that a more acute papillary muscle angle to the LAVV annulus was the only differentiator between those who developed LAVVR and those who did not (Figure 1). These contradicting reports on the contribution of subvalvular apparatus suggest further studies are warranted. In contrast, the lack of difference in leaflet size, degree of leaflet tethering, and prolapse are consistently reported, suggesting that these preoperative factors, albeit important in AVV function, may play a smaller role in determining postoperative LAVVR.
Figure 1: Quantitative 3DE Preoperative Features Associated With Postoperative LAVVR
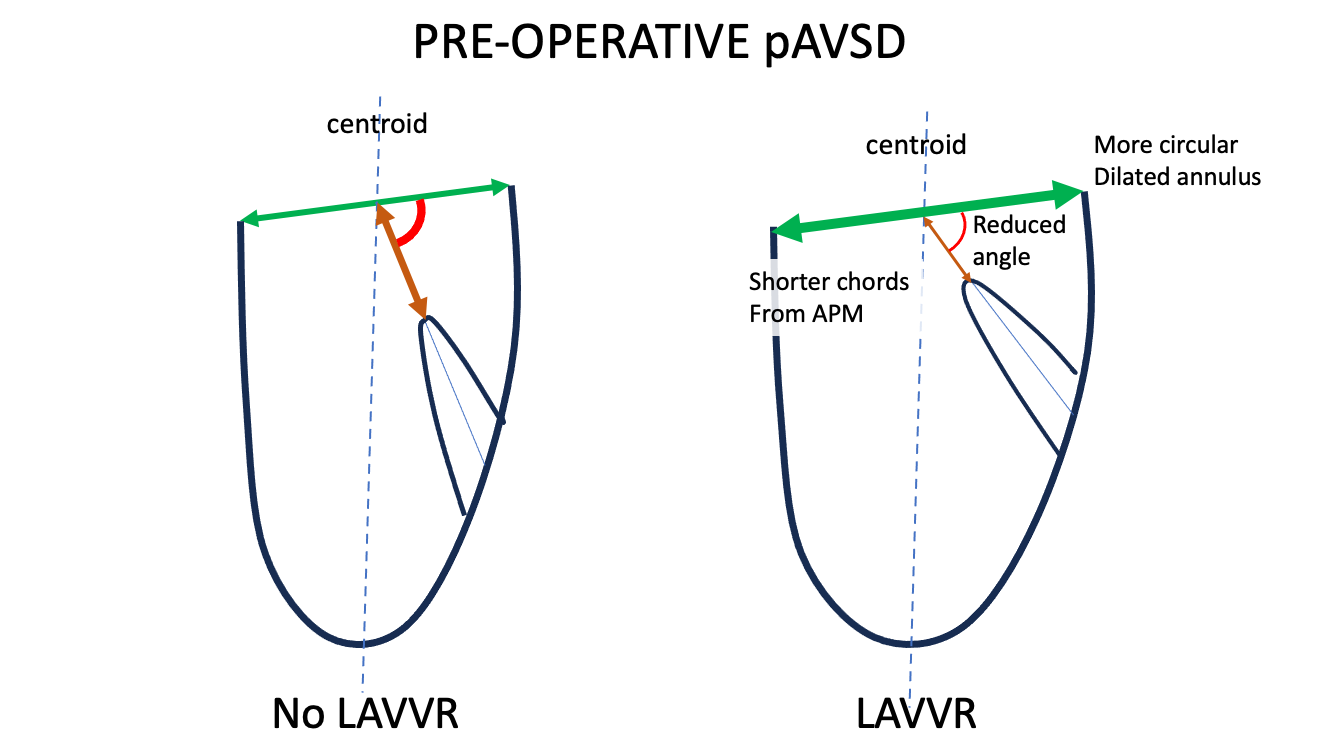
3DE = three-dimensional echocardiography; APM = anterior papillary muscle; LAVVR = left atrioventricular valve regurgitation; pAVSD = partial atrioventricular septal defect.
Currently, the clinical use of 3DE relies almost completely on qualitative assessment of the AVV. For more accurate and reliable prognostication of LAVV outcomes after surgery, routine quantitative assessment is necessary. This approach requires the development of highly automated and efficient quantitative tools that can segment the components of the valve unit. Accurate automatic segmentation of the tricuspid valve in the setting of hypoplastic left heart syndrome is now possible with adequate training of a deep learning algorithm with 3DE datasets.9 In addition, three-dimensional (3D) structure analysis progressively abandons conventional Euclidian geometry for nonplanar shape analysis to assess the structure as a complex 3D whole rather than the current approach of reducing the valve into two-dimensional planar objects for measurements.10 Nevertheless, the efforts to unravel the mystery of post LAVVR in AVSD continues; perhaps by taking the search into the third dimension, clinicians will fare better than their predecessors. There is much work to be done before clinicians will be able to collaborate with their surgical colleagues to explore newer surgical techniques, early postoperative care, and long-term outpatient medical management, and before it will be possible to mitigate the still-significant burden of morbidity and mortality from postoperative LAVVR and reoperation.
References
- Nathan M, Levine JC, Van Rompay MI, et al.; Pediatric Heart Network Investigators. Impact of major residual lesions on outcomes after surgery for congenital heart disease. J Am Coll Cardiol 2021;77:2382-94.
- Mery CM, Zea-Vera R, Chacon-Portillo MA, et al. Contemporary results after repair of partial and transitional atrioventricular septal defects. J Thorac Cardiovasc Surg 2019;157:1117-27.
- Fong LS, Betts K, Kannekanti R, Ayer J, Winlaw DS, Orr Y. Modified-single patch vs double patch repair of complete atrioventricular septal defects. Semin Thoracic Cardiovasc Surg 2020;32:108-116.
- Takahashi K, Mackie AS, Thompson R, et al. Quantitative real-time three-dimensional echocardiography provides new insight into the mechanisms of mitral valve regurgitation post-repair of atrioventricular septal defect. J Am Soc Echocardiogr 2012;25:1231-44.
- Colen TM, Khoo NS, Ross DB, Smallhorn JF. Partial zone of apposition closure in atrioventricular septal defect: are papillary muscles the clue. Ann Thorac Surg 2013;96:637-43.
- Ando M, Takahashi Y. Variations of atrioventricular septal defects predisposing to regurgitation and stenosis. Ann Thorac Surg 2010;90:614-21.
- Buratto E, Lui A, Hu T, et al. Propensity score matched analysis of cleft closure in complete atrioventricular septal defect repair. Ann Thorac Surg 2022;113:1553-62.
- Nam HH, Herz C, Lasso A, et al. Visualization and quantification of the unrepaired complete atrioventricular canal valve using open-source software. J Am Soc Echocardiogr 2022;35:985-96.
- Herz C, Pace DF, Nam HH, et al. Segmentation of tricuspid valve leaflets from transthoracic 3D echocardiograms of children with hypoplastic left heart syndrome using deep learning. Front Cardiovasc Med 2021;8:[ePub ahead of print].
- Amin S, Dewey H, Lasso A, et al. Euclidean and shape-based analysis of the dynamic mitral annulus in children using a novel open-source framework. J Am Soc Echocardiogr 2024;37:259-67.
Clinical Topics: Congenital Heart Disease and Pediatric Cardiology, Noninvasive Imaging, Congenital Heart Disease, CHD and Pediatrics and Imaging, Echocardiography/Ultrasound, Valvular Heart Disease
Keywords: Heart Septal Defects, Echocardiography, Echocardiography, Three-Dimensional, Heart Defects, Congenital