Emerging Trends in Multimodality Cardiovascular Imaging at AHA25
Quick Takes
- Artificial intelligence (AI)–supported imaging refines risk assessment in nonobstructive coronary artery disease (CAD).
- Cardiovascular magnetic resonance (CMR) improves diagnostic precision for angina with no obstructive CAD.
- Multimodal AI increases early detection of transthyretin cardiac amyloidosis.
Introduction
The American Heart Association (AHA) Scientific Sessions 2025 (AHA25) highlighted how quickly cardiovascular (CV) imaging is shifting toward integrated, biology-centered care. Across multiple presentations in New Orleans, investigators showed how advanced analytics applied to computed tomography (CT), cardiovascular magnetic resonance (CMR), echocardiography, and nuclear imaging can reveal early disease pathways and guide more individualized therapy. Several studies focused on nonobstructive coronary artery disease (CAD), a condition that often receives less attention despite a substantial burden of future CV events. Others demonstrated that artificial intelligence (AI)–supported approaches can shorten the diagnostic timeline for transthyretin cardiac amyloidosis (ATTR), improving opportunities for timely intervention.
Reframing Nonobstructive CAD
An important update at the meeting was the 2025 AHA scientific statement on the Evaluation and Medical Management of Nonobstructive CAD in Patients With Chest Pain, which emphasized that this condition should no longer be viewed as benign. Instead, quantitative imaging can be used to identify individuals at increased risk so that prevention strategies can be applied earlier. This framework sets the stage for many of the late-breaking studies.1
Advances in Coronary CT Angiography
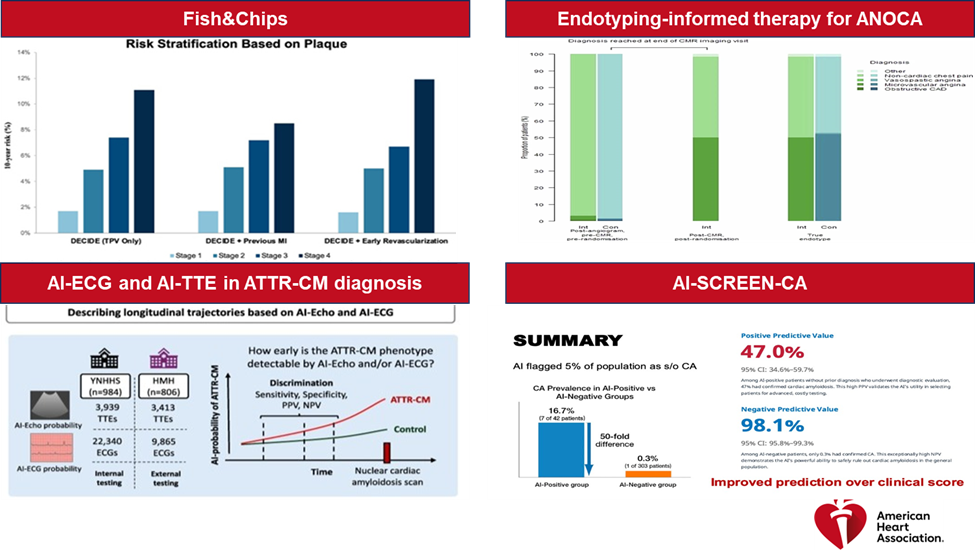
Coronary computed tomographic angiography (CCTA) continues to evolve by providing a more nuanced assessment of plaque morphology. New data from the Fish&Chips (FFRCT in stable heart disease and CCTA helps improve patient care and societal costs) study, led by Parsa et al., offered robust evidence that total plaque volume is a meaningful marker of long-term vulnerability.2 Using AI-based plaque quantification, the investigators showed that greater total plaque burden was closely associated with higher rates of future CV events. The relationship persisted regardless of stenosis severity, bolstering the claim that total plaque burden, rather than degree of stenosis, is the major risk predictor of outcomes.
A complementary analysis from the same research group examined a broad clinical cohort staged by predefined plaque volume thresholds. Event rates rose progressively across stages. Individuals in the earliest stage had the lowest rates of cardiac death or nonfatal myocardial infarction, whereas those in the most advanced stage experienced rates of >10%. The study also modeled preventive strategies guided by plaque burden and found that tailoring low-density lipoprotein cholesterol goals to plaque stage improved treatment efficiency compared with a uniform approach. These findings suggest that plaque burden may help clinicians deliver more personalized prevention by aligning therapy intensity with underlying disease activity.
A study using CONFIRM2 (Quantitative COroNary CT Angiography Evaluation For Evaluation of Clinical Outcomes: An InteRnational, Multicenter Registry) data focused on noncalcified plaque volume.3 AI-enhanced CT was used in this international cohort to compare total noncalcified plaque volume with the ratio of noncalcified to calcified plaque. Although both parameters showed unadjusted associations with clinical events, only noncalcified plaque volume remained a significant predictor after multivariable adjustment. These findings highlight that the total amount of biologically active, lipid-rich plaques are more influential than the proportional balance between plaque types. The results also underscore that coronary artery calcium scoring, although valuable, does not fully capture risk because individuals with minimal calcification may still harbor substantial noncalcified plaque.
Functional CMR for Diagnostic Clarification
In the CorCMR (Coronary Microvascular Angina Cardiac Magnetic Resonance Imaging) randomized trial, adenosine stress CMR–guided management was compared with usual care in individuals presenting with angina and no obstructive CAD.4 CMR reclassified the working diagnosis in more than one-half of participants, and microvascular angina emerged as the dominant endotype in the intervention arm. Only a small fraction of patients in the control arm received this diagnosis.
Symptom improvement closely paralleled the diagnostic clarity. After 12 months, significant improvement in Seattle Angina Questionnaire scores and health-related quality of life were observed in the CMR-guided group. These results reinforce the value of functional testing in uncovering microvascular dysfunction and directing therapy toward the underlying pathophysiology.
AI Strengthens Amyloidosis Detection
Several presentations showcased how AI can streamline the process of identifying ATTR. The AI-SCREEN-CA (Artificial Intelligence-based Software for Checking REal-world Echocardiography to ideNtify hidden Cardiac Amyloidosis) study by Sakamoto et al. used a deep learning model applied to routine echocardiograms to identify patients who might benefit from additional testing.5 With a negative predictive value >98%, the results suggest that AI-assisted echocardiography can serve as an efficient initial screen.
Jain et al. extended this concept by merging AI analysis of electrocardiographic (ECG), echocardiographic, and demographic data. This approach identified patients with ATTR with a positivity rate almost three times that of historical control patients.6
The TRACE-AI Network study by Oikonomou et al. evaluated an algorithm combining both ECG-derived and transthoracic echocardiography (TTE) AI–derived data points to help with ATTR cardiomyopathy diagnosis (confirmed by nuclear cardiac amyloid testing). Of the 656 participants, only 69 screened positive on both modalities (ECG and TTE), which resulted in a specificity of 0.93-0.94 and a reduction in false-positives compared with AI-ECG or AI-echocardiography alone.7
In another contribution, El Yaman et al. assessed the joint prognostic value of rubidium positron emission tomography–derived myocardial flow reserve and echocardiography-assessed diastolic dysfunction. There was a strong correlation between diastolic function and flow reserve. The combination of impaired myocardial flow reserve and advanced diastolic dysfunction was linked to the highest event rates, strengthening predictive performance beyond either metric alone.8
Clinical Implications
The findings presented at AHA25 illustrate a growing consensus that CV imaging must integrate biological insights, functional data, and AI-based analytics. CCTA can quantify disease activity long before plaque becomes obstructive. CMR can identify the endotype behind persistent angina. Echocardiography, ECG, and nuclear imaging can detect ATTR with greater accuracy when supported by multimodal AI.
These developments point toward more individualized CV care (Figure 1). Imaging can help clinicians select therapies according to disease biology rather than relying solely on anatomical findings, which may improve outcomes for patients whose conditions have historically been underrecognized.
Figure 1: Findings From Select Studies Presented at AHA252,4,5,7
AHA25 = American Heart Association Scientific Sessions 2025.
References
- Slipczuk L, Blankstein R, Bucciarelli-Ducci C, et al. State of the art: evaluation and medical management of nonobstructive coronary artery disease in patients with chest pain: a scientific statement from the American Heart Association. Circulation. 2025;152(23):e443-e466. doi:10.1161/CIR.0000000000001394
- Parsa S, Peng A, Fairbairn T, et al. Abstract 4335849: Improvements in Diagnostic and Therapeutic Cardiovascular Risk Assessment Through Total Plaque Volume Burden: An Analysis of the Fish&Chips Study (AHA Journals website). 2025. Available at: https://www.ahajournals.org/doi/10.1161/circ.152.suppl_3.4335849. Accessed 02/09/2026.
- van Rosendael A, al-Mallah M, Alasnag M, et al. 4393099: Risk stratification by AI-guided CT coronary atherosclerosis quantification alongside the clinical likelihood of obstructive stenosis (AHA Journals website). 2025. Available at:. https://www.ahajournals.org/doi/10.1161/CIR.0000000000001403. Accessed 01/07/2026.
- Bradley CP, McKinley G, Orchard V, et al. Endotyping-informed therapy for patients with chest pain and no obstructive coronary artery disease: a randomized trial. Nat Med. 2026;32(1):332-341. doi:10.1038/s41591-025-04044-4
- Sakamoto A, Kagiyama N, Nakamura Y, et al. Artificial Intelligence-based Software for Checking REal-world Echocardiography to ideNtify hidden Cardiac Amyloidosis: AI-SCREEN-CA. Presented at the American Heart Association Scientific Sessions 2025, New Orleans, LA. November 10, 2025.
- Jain SS, Sun T, Pierson E, et al. Detecting transthyretin cardiac amyloidosis with artificial intelligence: a nonrandomized clinical trial. JAMA Cardiol. Published online November 10, 2025. doi:10.1001/jamacardio.2025.4591
- Oikonomou EK, Sangha V, Shankar SV, et al. Artificial intelligence-enabled electrocardiography and echocardiography to track preclinical progression of transthyretin amyloid cardiomyopathy. Eur Heart J. 2025;46(37):3651-3662. doi:10.1093/eurheartj/ehaf450
- El Yaman A, Sayed A, Alwan M, et al. Abstract 4367325: Correlation and Complementary Prognostic Value of 82Rb PET-Derived Myocardial Flow Reserve and Echocardiography-Assessed Diastolic Dysfunction (AHA/ASA Journals website). 2025. Available at: https://www.ahajournals.org/doi/10.1161/circ.152.suppl_3.4367325. Accessed 01/12/2026.
Clinical Topics: Cardiovascular Care Team, Invasive Cardiovascular Angiography and Intervention, Noninvasive Imaging, Interventions and Imaging, Computed Tomography, Echocardiography/Ultrasound, Magnetic Resonance Imaging, Nuclear Imaging, Diabetes and Cardiometabolic Disease, Dyslipidemia, Heart Failure and Cardiomyopathies, Stable Ischemic Heart Disease, Prevention, Valvular Heart Disease
Keywords: Imaging, Multimodal Imaging, Computed Tomography Angiography, Magnetic Resonance Imaging, Echocardiography, Positron-Emission Tomography, Artificial Intelligence, AHA25