Cutting-Edge Structural Interventions | Tricuspid Regurgitation: When Is Intervention Indicated?

Management of tricuspid regurgitation (TR) largely depends on the etiology, which falls under three categories: 1) primary TR (due to anatomic abnormalities of the tricuspid valve (TV), 2) secondary TR (mostly due to left-sided pathology producing pulmonary hypertension [PH] and right ventricular [RV] dysfunction), and 3) right-sided cardiac implantable electronic device (CIED)-related TR (e.g., transvenous pacemakers).1-3 Primary TR accounts for only about 10% of TR with secondary TR and CIED-related TR accounting for the rest.1-3
First steps in management of TR should address the underlying causes of heart failure symptoms.4 For patients presenting with mild to moderate TR, an appropriate diuretic regimen for volume management may achieve symptom resolution. However, there are many patients with persistent severe TR despite these measures and TV intervention should be considered when patients remain symptomatic or develop evidence of RV dysfunction or dilation.1-3
Tricuspid intervention for TR has historically been confined to surgical approaches. Isolated TV surgery is associated with high rates of in-hospital mortality.1,2 While new risk models have provided more clarity on appropriate patient selection for TV surgery, transcatheter tricuspid valve interventions (TTVI) have emerged to provide additional treatment options for patients with severe TR.1-5 The most common percutaneous devices involve either tricuspid transcatheter edge-to-edge repair (T-TEER) or transcatheter tricuspid valve replacement (TTVR).6-14
Two pivotal trials, TRILUMINATE and TRISCEND II, respectively compared T-TEER using Triclip (Abbott Vascular, Santa Clara, CA) to medical therapy and TTVR using Evoque (Edwards Lifesciences, Irvine, CA) to medical therapy in patients with symptomatic severe TR.10,11 While neither trial demonstrated a mortality benefit, these two trials showed that both T-TEER and TTVR were associated with a reduction in TR, reduction of symptoms and improvement in quality of life compared to medical therapy alone.
It should be noted that the patients enrolled in these trials were carefully selected. More specifically, they had TV anatomy suitable to T-TEER or TTVR and were not being considered for TV surgery because of their high surgical risk.10,11 Furthermore, these patients did not have severe left ventricular (LV) dysfunction, severe PH (pulmonary artery systolic pressures >70 mm Hg), severe RV dysfunction, TV stenosis, additional valve dysfunction requiring intervention, or prior tricuspid interventions (TV replacements or repair) that would interfere with placement of the T-TEER or TTVR device. Additionally, TTVI trials excluded end-stage renal disease (ESRD) patients, which is an important demographic given the prevalence of TR in these patients. Altogether, there are no trials to date suggesting that patients with any of these exclusion criteria will benefit from T-TEER or TTVR. Valve-in-valve and valve-in-ring TTVRs have been studied, but this typically involves off-label use of Sapien 3 (Edwards Lifesciences) valves or Melody (Medtronic, Minneapolis, MN) valves as opposed to Triclip or Evoque devices.15 While patients with CIED leads frequently undergo TTVI, the presence of CIED leads requires careful consideration as the CIED lead may be causing CIED-related TR that potentially can improve with lead extraction or repositioning.2,3,16,17 However, these CIED lead interventions could themselves damage the tricuspid valve.2,3,16,17 All of these clinical points need to be considered when considering TTVI (Figure 1).
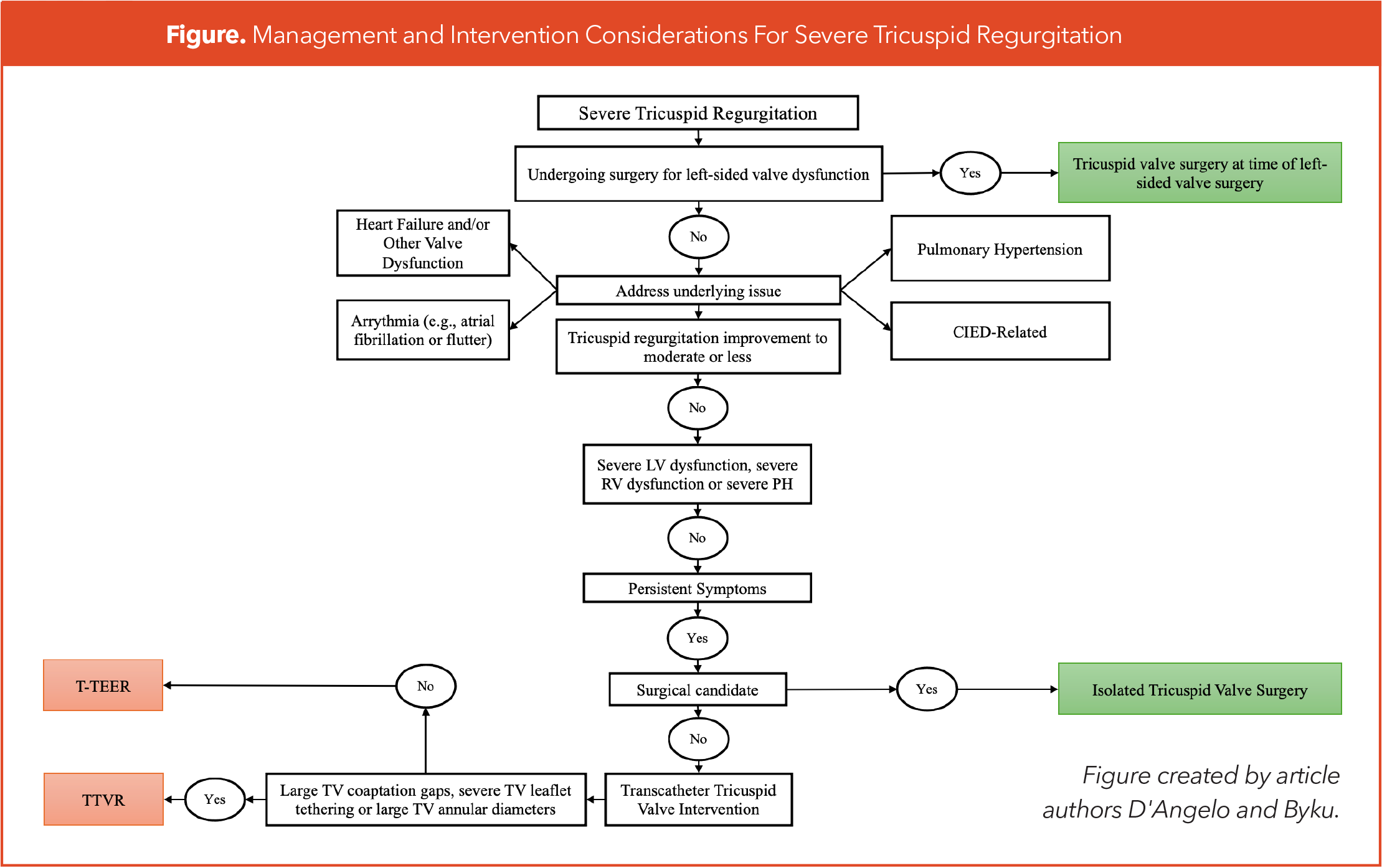
Once a decision has been made to pursue TTVI, a choice between T-TEER and TTVR should be made. T-TEER does not reduce TR severity as much as TTVR and has more anatomic restrictions including the presence of large TV coaptation gaps (>10 mm), severe TV leaflet tethering and large TV annular diameters. However, TTVR's more complete resolution of TR may cause worsening right-sided heart failure in some patients and the more modest TR reduction seen with T-TEER may be better tolerated in those with some degree of RV dysfunction.
Furthermore, T-TEER is a simpler procedure with a lower adverse event rate including less bleeding, less interaction with existing CIEDs and fewer conduction abnormalities necessitating permanent pacemaker placement.2,3,18 In patients with pre-existing CIEDs, TTVR is considered more favorable in the setting of CIED-related TR. However, TTVR may cause lead jailing and T-TEER may be more favorable when the CIED leads are far away from the main TR jet location.3 In light of all this, TTVI planning in the setting of CIED leads should involve a multidisciplinary team involving the structural interventionalist, structural imagers and electrophysiologists.15 Additionally, TTVR requires pre-procedure CT scanning and post-procedure anticoagulation, which is less desirable in patients with pre-existing kidney dysfunction and higher risks of bleeding, respectively.2,3,18
TTVI has emerged as a promising treatment option for select patients with symptomatic severe TR, but there remains limited data on TTVI within important patient cohorts. Commercial liberalization of the Evoque and Triclip devices will hopefully provide more answers, and further studies are needed to better identify which patients will benefit the most from TTVI. For now, a multidisciplinary approach will continue to be at the center of decision making.

References
- Otto CM, Nishimura RA, Bonow RO, et al. 2020 ACC/AHA guideline for the management of patients with valvular heart disease: A report of the American College of Cardiology/American Heart Association Joint Committee on Clinical Practice Guidelines. JACC. 2021;77(4):e25-e197. doi/10.1016/j.jacc.2020.11.018
- Praz F, Borger MA, Lanz J, et al. 2025 ESC/EACTS guidelines for the management of valvular heart disease. Eur Heart J. 2025;46(44):4635-4736. doi:10.1093/eurheartj/ehaf194
- O'Gara PT, Lindenfeld J, Hahn RT, et al. 10 issues for the clinician in tricuspid regurgitation evaluation and management: 2025 ACC Expert Consensus Decision Pathway: A Report of the American College of Cardiology Solution Set Oversight Committee. JACC. 2026;87(4):447-86. doi:10.1016/j.jacc.2025.07.002
- Adamo M, Chioncel O, Pagnesi M, et al. Epidemiology, pathophysiology, diagnosis and management of chronic right-sided heart failure and tricuspid regurgitation. A clinical consensus statement of the Heart Failure Association (HFA) and the European Association of Percutaneous Cardiovascular Interventions (EAPCI) of the ESC. Eur J Heart Fail. 2024;26(1):18-33. doi:10.1002/ejhf.3106
- Dreyfus J, Audureau E, Bohbot Y, et al. TRI-SCORE: a new risk score for in-hospital mortality prediction after isolated tricuspid valve surgery. Eur Heart J. 2022;43(7):654-662. doi:10.1093/eurheartj/ehab679
- Taramasso M, Benfari G, van der Bijl P, et al. Transcatheter versus medical treatment of patients with symptomatic severe tricuspid regurgitation. JACC. 2019;74(24):2998-3008. doi:10.1016/j.jacc.2019.09.028
- Perlman GY, Dvir D. Treatment of tricuspid regurgitation with the FORMA repair system. Front Cardiovasc Med. 2018;5:140. doi:10.3389/fcvm.2018.00140
- Kodali SK, Hahn RT, Davidson CJ, et al. 1-Year outcomes of transcatheter tricuspid valve repair. JACC. 2023;81(18):1766-1776. doi:10.1016/j.jacc.2023.02.049
- Kodali S, Hahn RT, Makkar R, et al. Transfemoral tricuspid valve replacement and one-year outcomes: the TRISCEND study. Eur Heart J. 2023;44(46):4862-4873. doi:10.1093/eurheartj/ehad667
- Sorajja P, Whisenant B, Hamid N, et al. Transcatheter repair for patients with tricuspid regurgitation. N Engl J Med. 2023;388(20):1833-1842. doi:10.1056/NEJMoa2300525
- Hahn RT, Makkar R, Thourani VH, et al. Transcatheter valve replacement in severe tricuspid regurgitation. N Engl J Med. 2025;392(2):115-126. doi:10.1056/NEJMoa2401918
- Tang GHL, Hahn RT, Whisenant BK, et al. Tricuspid transcatheter edge-to-edge repair for severe tricuspid regurgitation: 1-Year outcomes from the TRILUMINATE randomized cohort. JACC. 2025;85(3):235-246. doi:10.1016/j.jacc.2024.10.086
- Rogers T, Ratnayaka K, Sonmez M, et al. Transatrial intrapericardial tricuspid annuloplasty. JACC Cardiovasc Interv. 2015;8(3):483-491. doi:10.1016/j.jcin.2014.10.013
- Lurz P, Kresoja KP, Besler C, et al. Heterotopic crosscaval transcatheter tricuspid valve replacement for patients with tricuspid regurgitation: The Trillium device. JACC Cardiovasc Interv. 2025;18(11):1425-1434. doi:10.1016/j.jcin.2025.04.036
- Sanon S, Cabalka AK, Babaliaros V, et al. Transcatheter tricuspid valve-in-valve and valve-in-ring implantation for degenerated surgical prosthesis. JACC Cardiovasc Interv. 2019;12(15):1403-1412. doi:10.1016/j.jcin.2019.05.029
- Hahn RT, Wilkoff BL, Kodali S, et al. Managing implanted cardiac electronic devices in patients with severe tricuspid regurgitation: JACC state-of-the-art review. JACC. 2024;83(20):2002-2014. doi:10.1016/j.jacc.2024.02.045
- Park SJ, Gentry JL 3rd, Varma N, et al. Transvenous extraction of pacemaker and defibrillator leads and the risk of tricuspid valve regurgitation. JACC Clin Electrophysiol. 2018;4(11):1421-1428. doi:10.1016/j.jacep.2018.07.011
- Kempton H, Stolz L, Weckbach L, et al. Choosing between tricuspid repair or replacement: decision algorithms. Struct Heart. 2025;9(11):100732. Published 2025 Sep 30. doi:10.1016/j.shj.2025.100732
Keywords: Cardiology Magazine, ACC Publications, CM-Apr-2026, Tricuspid Valve, Regurgitation, Structural Intervention
