Cutting-Edge Structural Interventions | Transcatheter Electrosurgery: Advancing Treatment Horizons
Transcatheter electrosurgery is a technique that has evolved during the ongoing journey to find sophisticated strategies for patients with structural heart disease who are otherwise untreatable. This new technique involves delivering continuous alternating high-frequency current (radiofrequency [RF]) to a specific target via an insulated guidewire. The guidewire is attached to a generator enabling the operator to vaporize the tissue and either traverse or lacerate it.1
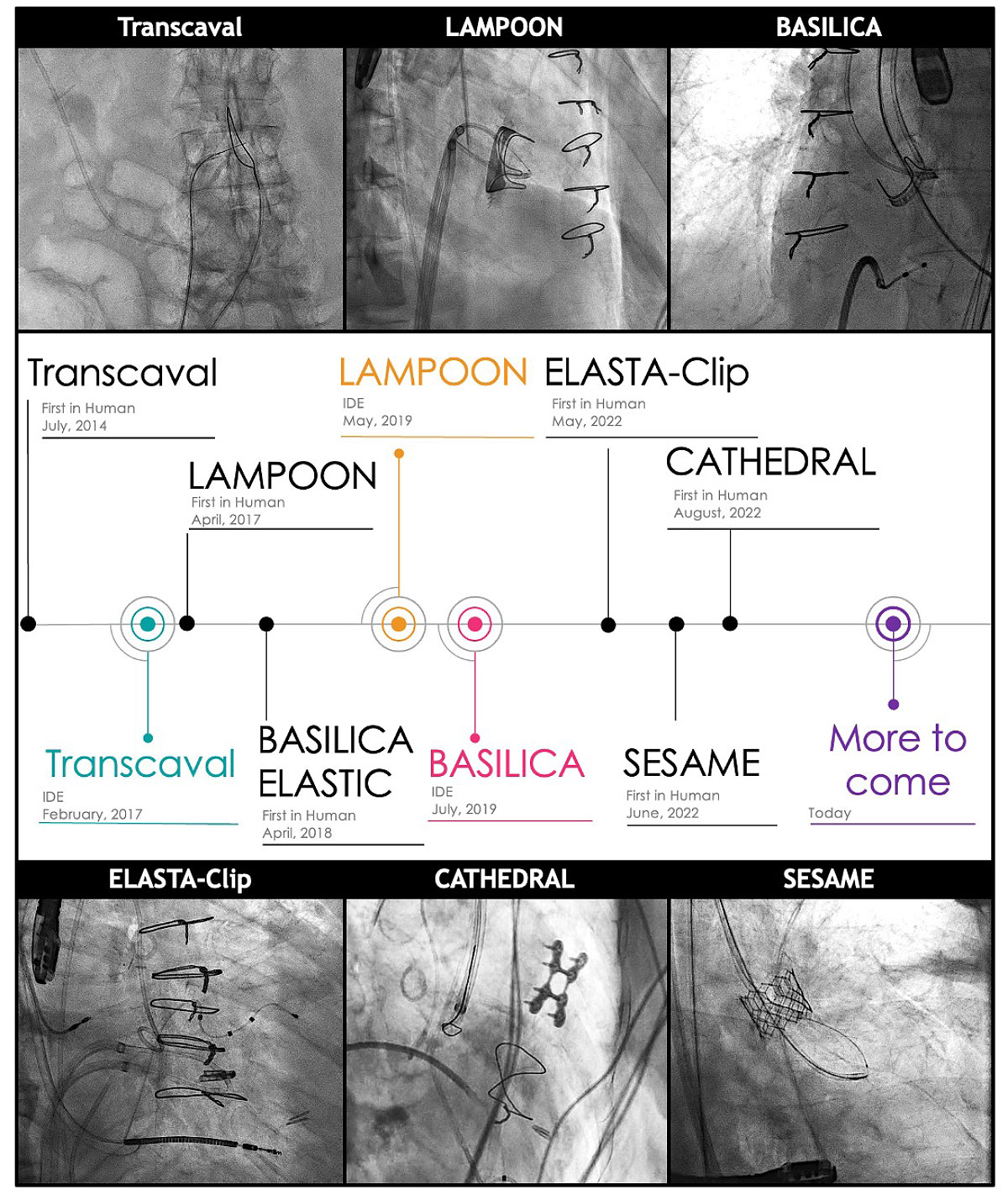
Transcatheter electrosurgery has been applied in three broad categories: 1) traversal of occluded lesions (pulmonary valve atresia; chronic total occlusion of arterial and venous system), 2) traversal between two cardiovascular structures (transseptal puncture; transcaval access), and 3) laceration (LAMPOON, BASILICA, CATHEDRAL, ELASTIC, ELASTA-Clip, SESAME). The role of this novel group of therapeutic procedures has quickly increased in the past decade and continues to grow. Here we review its chronological history, summarize each technique, and introduce more recent additions to the family of transcatheter electrosurgery therapy.
Traversal of Occluded Lesions
One of the first applications of electrosurgery during a transcatheter procedure was reported in 1993, in which an RF-energized guidewire was used to penetrate an atretic pulmonary valve in a child. A dilation balloon was then placed through the perforated atretic tunnel to open the pulmonary outflow tract.2 Numerous case reports followed, showing promising success rates and relatively low procedural complications.3
Since then, recanalizing occluded structures using an RF energized guidewire is now commonly used in both central and peripheral vascular interventions. These have included recanalization of pulmonary artery branch occlusions,4 longstanding subclavian vein occlusions,5 complete aortic coarctation,6 and chronic coronary total occlusions.7 Most of these cases used RF puncture as an adjunct to conventional approaches, and thus were used relatively sporadically.
Traversal Between Cardiovascular Structures
Transseptal Puncture
A more systematic application of RF puncture in structural heart intervention was first used for transseptal puncture.8 The conventional Brockenbrough needle puncture approach has important limitations, including requirements of force to puncture, unpredictable movement of the needle following the puncture with risk of subsequent perforation of the left atrium, and difficulty crossing a highly compliant, aneurysmal, or calcified septum. RF transseptal puncture using a dedicated RF needle (NRG Transseptal Needle, Baylis Medical, Montreal, Canada) allows atrial septal traversal without relying on mechanical force. This provides a more reliable puncture, especially in a septum that is elastic or aneurysmal and minimizes the risk of left atrial perforation.
A randomized controlled trial comparing RF vs. conventional transseptal left atrial access showed a significantly higher success rate (100% vs. 72.2%) and a shorter puncture time (2.3 vs. 7.3 minutes) using RF transseptal puncture.9
Transcaval Access
Transcaval access is a strategy to access the descending aorta via a femoral vein puncture in patients with severe iliofemoral arterial disease. For example, it is now used in selected patients for transcatheter aortic valve replacement (TAVR) and mechanical ventricular assist device insertion.
Through transfemoral venous access, a stiff guidewire (typically Astato XS 20, Asahi Intecc, Aichi, Japan), insulated in a microcatheter (typically Piggyback Wire Converter, Teleflex, Morrisville, NC) is advanced into the inferior vena cava (IVC). At a predetermined crossing plane with no calcium deposition (determined by careful preprocedural CT analysis), the guidewire is electrified and advanced from the IVC through the short retroperitoneal space into the abdominal aorta.
A snare previously placed in the descending aorta from the femoral artery is used to capture the guidewire tip in the aorta and advance the guidewire into the thoracic aorta. A large-bore sheath is then advanced over the wire guide from the femoral vein into the aorta for completion of the procedure.
At the end of the procedure, the large-bore sheath is removed and the aorto-IVC tunnel is closed using a nitinol cardiac occluder (Amplatzer Duct Occluder or Amplatzer Septal Defect Occluder, Abbott, Abbott Park, IL; or Transcaval Closure Device, Transmural System, Boston, MA). Retroperitoneal bleeding during closure is minimized because retroperitoneal pressure is higher than the venous pressure and the blood flows directly from left to right into the IVC.
A prospective trial enrolling 100 patients demonstrated a 99% success rate. Surprisingly, major vascular complications (mostly retroperitoneal hematoma) occurred in only 13% and there was only one death.10, 11 A recent comparison of transcaval access with transaxillary access TAVR showed a five-fold lower incidence of stroke (2.5% vs. 13.2%) with similar bleeding, vascular complications and survival.12 Furthermore, operator radiation exposure and ergonomics were the same as for transfemoral arterial access TAVR.
Laceration
An evolution from mere "puncture" to "laceration" has revolutionized the field of transcatheter electrosurgery, bringing opportunities for a variety of adjunctive procedures to replace surgical intervention.
To allow laceration of tissue, RF energy has to be directed along a straight line. This is accomplished by focally denuding insulation of a steel guidewire (Astato XS 20) over an approximately 2 mm length near its center. A kink ("flying V") is then made at the middle of the denuded portion to keep the denuded portion from moving. RF energy is then applied to the tissue as the wire is pulled back by both ends, cutting the tissue.
LAMPOON
Intentional laceration of the anterior mitral leaflet to prevent outflow obstruction (LAMPOON) was the first of a series of RF lacerations of native or bioprosthetic tissues. LAMPOON is performed immediately preceding transcatheter mitral valve replacement (TMVR) in patients at risk of left ventricular outflow tract (LVOT) obstruction by the residual anterior mitral leaflet when the TMVR is completed.
Using the retrograde aortic approach across the aortic valve, the anterior mitral leaflet is perforated at its base using a stiff guidewire through a coronary guide catheter. The guidewire is then advanced to the apex of the left ventricle and snared. A "flying V" is prepared in the midportion of the guidewire outside the body.
The proximal portion of the guidewire is advanced and the snared distal portion is pulled back simultaneously until the "flying V" is in position across the perforated base of the mitral valve. RF energy is applied while infusing nonionic 5% dextrose so as to confine RF energy to the targeted tissue and avoid clotting. Thus, a split from the base of the mitral leaflet to its tip is produced. Blood now can pass through the LVOT via the split which avoids LVOT obstruction after TMVR.
Several iterations of this procedure have been developed, but all produce the same anterior mitral laceration.13 Patients with preexisting annuloplasty rings or surgical mitral valves can also undergo tip-to-base LAMPOON if they are at risk of LVOT obstruction.14 LAMPOON and subsequent TMVR was successful in 100% of the patients included in the investigational device exemption (IDE) trial, with 93% survival to discharge and 30 days.14 LVOT gradient <30 mm Hg was achieved in 97% after TMVR.
BASILICA
Bioprosthetic or native aortic scallop intentional laceration to prevent artery obstruction (BASILICA) is used in patients at risk for coronary artery obstruction mostly due to aortic valve leaflets which could occlude the coronary ostium after native or valve-in-valve TAVR.
Built upon techniques used in LAMPOON, a native or bioprosthetic leaflet overlying the threatened coronary ostium (of either the left or right coronary) is traversed at its base from above the aortic annulus and ensnared in the LVOT. The "flying V" is advanced to the base of the aortic leaflet while the distal tip is simultaneously pulled back. The wire is energized while pulling both ends, allowing the "flying V" to lacerate the leaflet. This preserves coronary artery flow through the leaflet split into the coronary ostium after the TAVR procedure.
In a real-world study, 214 patients from 25 North American and European centers with risk of coronary occlusion after TAVR had a 94.4% success rate. The 30-day mortality and stroke rate remained low (2.8% and 2.8%, respectively).15 Data at one year from the IDE trial showed no additional strokes or myocardial infarctions after 30 days.16
CATHEDRAL
Leaflet laceration alone may not always resolve the problem of threatened coronary obstruction with low lying coronaries. Despite BASILICA some patient with prolapsed or elongated aortic leaflets may not avoid the risk of coronary artery obstruction following TAVR. Catheter electrosurgical debulking and removal (CATHEDRAL) which totally removes the offending bioprosthetic leaflet has been described in a case report.17
Using the same techniques as in BASILICA, the base of the leaflet at risk is traversed, and a "flying V" is advanced. Unlike BASILICA, the "flying V" is not focally denuded or energized for laceration. Instead a loop snare is advanced over the "flying V" and grasps the entire leaflet. Tension is applied and the loop snare is energized. The leaflet is therefore detached from the aortic annulus and removed.
ELASTIC and ELASTA-Clip
Patients with a failed Alfieri stitch or a failed MitraClip present a difficult clinical problem. Most are not good surgical candidates. The strategy to place a transcatheter mitral valve by reopening the mitral orifice can be achieved by using transcatheter electrosurgery followed immediately by TMVR.
The so-called ELASTIC and ELASTA-Clip procedures detach the stitch/clip from the anterior mitral leaflet that enables TMVR through the enlarged single orifice. In short, a loop is created with a wire guide from a transeptal approach through the double orifice. A "flying V" is advanced so that it is positioned in front of the stitch/clip in the anterior mitral leaflet. RF energy is applied and the stitch/clip is separated from the anterior mitral leaflet and remains attached to the posterior leaflet. TMVR is then immediately performed in the single mitral orifice.
The procedure has been reported in one case of failed Alfieri stitch, a case series of six patients with failed MitraClip with subsequent investigational Tendyne valve (Abbott) implantation, and one patient with failed MitraClip with a subsequent investigational M3 valve (Edwards Lifesciences, Irvine, CA) implantation, each enabling successful TMVR.18-20 As MitraClip is increasingly used, the number of patients with failed clips needing subsequent TMVR is expected to grow. One can easily foresee the future of ELASTA-Clip frequently being used in this setting.
SESAME
The latest addition to this series of electrosurgical technique is called SESAME (Septal scoring along the midline endocardium). SESAME produces septal reduction which mimics surgical left ventricular myotomy historically performed to treat hypertrophic cardiomyopathy. Since it performs LVOT myotomy, it can also be used to avoid LVOT obstruction after TMVR.
Using a retrograde transaortic approach, a guiding catheter tip is placed at the basal ventricular septum. Through it a stiff guidewire (typically Astato XS 20) is advanced along a predefined trajectory parallel to the septum until it reenters the left ventricular cavity approximately 2-3 cm below the insertion site. The guidewire tip is ensnared and externalized and the "flying V" is advanced to the distal septal/left ventricular cavity border. The "flying V" is energized and pulled back, splitting/splaying the myocardium from the mid-septum to the basal-septum which alleviates or prevents LVOT obstruction.
First-in-human experience has been reported in a patient at risk of LVOT obstruction resulting in a successful laceration, increase in LVOT measurements, and successful TMVR.21 A case series is currently underway and the results are promising to date. SESAME may become especially useful in patients with hypertrophic cardiomyopathy where transcatheter septal ablation with alcohol may not be successful due to coronary anatomy.
Advances Helping More Complex Patients
Electrosurgery techniques have advanced remarkably in the past decade, evolving from simple wire perforation and crossing anatomic structures to leaflet laceration, complete leaflet removal and myocardial laceration. A continuous and creative modification and optimization of these electrosurgery techniques by structural intervention experts has enabled treatment of the many complex patients, often deemed to be "patients with no options." The door is now opened wide for the next level of structural intervention.


This article was authored by Hiroki Ueyama, MD, and Isida Byku, MD, FACC, from the Division of Cardiology, Emory Structural Heart and Valve Center, Emory University Hospital Midtown, in Atlanta, GA.
References
- Khan JM, Rogers T, Greenbaum AB, et al. Transcatheter electrosurgery: JACC state-of-the-art review. J Am Coll Cardiol 2020;75:1455-70.
- Rosenthal E, Qureshi SA, Chan KC, et al. Radiofrequency-assisted balloon dilatation in patients with pulmonary valve atresia and an intact ventricular septum. Br Heart J 1993;69:347-51.
- Veldtman GR, Hartley A, Visram N, Benson LN. Radiofrequency applications in congenital heart disease. Expert Rev Cardiovasc Ther 2004;2:117-26.
- Fink C, Peuster M, Bertram H, Hausdorf G. Transcatheter recanalization of the left main pulmonary artery after four years of complete occlusion. Catheter Cardiovasc Interv 2001;53:81-4.
- Baerlocher MO, Asch MR, Myers A. Successful recanalization of a longstanding complete left subclavian vein occlusion by radiofrequency perforation with use of a radiofrequency guide wire. J Vasc Interv Radiol 2006;17:1703-6.
- Almashham Y, Dahdah N, Miro J. Use of radiofrequency then stent implantation for recanalization of complete aorta coarctation. Pediatr Cardiol 2008;29:207-9.
- Baim DS, Braden G, Heuser R, et al. Utility of the safe-cross-guided radiofrequency total occlusion crossing system in chronic coronary total occlusions (results from the Guided Radio Frequency Energy Ablation of Total Occlusions Registry Study). Am J Cardiol 2004;94:853-8.
- Sakata Y, Feldman T. Transcatheter creation of atrial septal perforation using a radiofrequency transseptal system: novel approach as an alternative to transseptal needle puncture. Catheter Cardiovasc Interv 2005;64:327-32.
- Hsu JC, Badhwar N, Gerstenfeld EP, et al. Randomized trial of conventional transseptal needle versus radiofrequency energy needle puncture for left atrial access (the TRAVERSE-LA Study). J Am Heart Assoc 2013;2:e000428.
- Greenbaum AB, Babaliaros VC, Chen MY, et al. Transcaval access and closure for transcatheter aortic valve replacement: A prospective investigation. J Am Coll Cardiol 2017;69:511-21.
- Lederman RJ, Babaliaros VC, Rogers T, et al. The fate of transcaval access tracts: 12-month results of the prospective NHLBI Transcaval transcatheter aortic valve replacement study. JACC Cardiovasc Interv 2019;12:448-56.
- Lederman RJ, Babaliaros VC, Lisko JC, et al. Transcaval versus transaxillary tavr in contemporary practice: A propensity-weighted analysis. JACC Cardiovasc Interv 2022;15:965-75.
- Lisko JC, Greenbaum AB, Khan JM, et al. Antegrade Intentional laceration of the anterior mitral leaflet to prevent left ventricular outflow tract obstruction: A simplified technique from bench to bedside. Circ Cardiovasc Interv 2020;13:e008903.
- Lisko JC, Babaliaros VC, Khan JM, et al. Tip-to-base LAMPOON for Transcatheter mitral valve replacement with a protected mitral annulus. JACC Cardiovasc Interv 2021;14:541-50.
- Khan JM, Babaliaros VC, Greenbaum AB, et al. Preventing coronary obstruction during transcatheter aortic valve replacement: results from the multicenter international BASILICA registry. JACC Cardiovasc Interv 2021;14:941-48.
- Khan JM, Greenbaum AB, Babaliaros VC ,et al. BASILICA trial: One-year outcomes of transcatheter electrosurgical leaflet laceration to prevent TAVR coronary obstruction. Circ Cardiovasc Interv 2021;14:e010238.
- Babaliaros VC, Gleason PT, Xie JX, et al. Toward transcatheter leaflet removal with the CATHEDRAL procedure: CATHeter Electrosurgical Debulking and RemovAL. JACC Cardiovasc Interv 2022;15:1678-1680.
- Khan JM, Lederman RJ, Sanon S, et al. Transcatheter mitral valve replacement after transcatheter electrosurgical laceration of alfieri STItCh (ELASTIC): First-in-human report. JACC Cardiovasc Interv 2018;11:808-11.
- Lisko JC, Greenbaum AB, Guyton RA, et al. Electrosurgical detachment of MitraClips from the anterior mitral leaflet prior to transcatheter mitral valve implantation. JACC Cardiovasc Interv 2020;13:2361-70.
- Inci EK, Greenbaum AB, Lederman RJ, et al. Transcatheter electrosurgical laceration and stabilization of failed MitraClip[s]/SAPIEN M3 for treatment of failed MitraClip. Circulation: Cardiovasc Interv 2022;15:e012014.
- Greenbaum AB, Khan JM, Bruce CG, et al. Transcatheter myotomy to treat hypertrophic cardiomyopathy and enable transcatheter mitral valve replacement: First-in-human report of septal scoring along the midline endocardium. Circ Cardiovasc Interv 2022;15:e012106.
Clinical Topics: Cardiac Surgery, Cardiovascular Care Team, Congenital Heart Disease and Pediatric Cardiology, Heart Failure and Cardiomyopathies, Invasive Cardiovascular Angiography and Intervention, Valvular Heart Disease, Aortic Surgery, Cardiac Surgery and CHD and Pediatrics, Cardiac Surgery and Heart Failure, Cardiac Surgery and VHD, Congenital Heart Disease, CHD and Pediatrics and Interventions, CHD and Pediatrics and Prevention, CHD and Pediatrics and Quality Improvement, Interventions and Structural Heart Disease, Mitral Regurgitation
Keywords: ACC Publications, Cardiology Magazine, Subclavian Vein, Sesamum, Transcatheter Aortic Valve Replacement, Heart Valve Diseases, Lacerations, Incidence, Electrosurgery, Patient Discharge, Prospective Studies, Dilatation, Punctures, Heart Defects, Congenital, Aorta, Catheters, Surgical Instruments, Ergonomics, Mitral Valve, Cytoreduction Surgical Procedures, Mitral Valve Insufficiency, Ventricular Outflow Obstruction, Endocardium, Cardiomyopathy, Hypertrophic, Myocardium, Heart Atria, Myotomy, Pectinidae, Glucose
< Back to Listings